Graziella Greco1, Joshua B. Slee1*
1 2755 Station Avenue, Department of Biology, DeSales University, Center Valley, PA 18034
* Joshua.Slee@Desales.edu
Abstract
Inflammatory diseases, such as cardiovascular disease (CVD), are significant concerns to the medical community and the health of societies worldwide. Strategies to reduce the inflammation associated with these diseases are of increased importance given the serious nature of CVD treatments, which are often risky and invasive. Some naturally occurring substances represent an untapped resource for potentially therapeutic compounds that may possess anti-inflammatory characteristics, such as resveratrol, a polyphenol-derived compound found in a variety of fruits. A goal of the study is to mimic an inflammatory response by inducing cell stress using Tumor Necrosis Factor-α (TNF-α) in vascular endothelial cells in vitro. The endothelial cells were treated with resveratrol to assess the anti-inflammatory properties of the compound. Resveratrol was shown to reduce cell stress without causing morphological abnormalities. In addition, wound healing, a critical step in the immune response to injury involving inflammation, was modeled using a scratch wound assay. Wound healing was enhanced in vascular endothelial cells. Lastly, we use polymeric biomaterials and a Chandler Loop to model resveratrol’s use in biomaterial rejection, another process highly dependent on inflammation levels. Resveratrol was shown to attenuate biomaterial rejection through significant inhibition (p < 0.01) of inflammatory cell attachment to polymeric biomaterials, including in the physiologically relevant model of a Chandler Loop. Taken together, our results suggest that resveratrol possesses potent anti-inflammatory characteristics in vascular endothelial cells and macrophages. These results take the field a step further in the prospect of using resveratrol for its anti-inflammatory properties and to increase the lifespan of implantable medical devices.
Introduction
In developed countries, such as the United States (US), cardiovascular disease (CVD) is often the leading cause of death, causing about 20% of deaths in the US between March 2020 and October 2021 (Shiels et al. 2022). The prevalence of CVD is likely linked to lifestyle habits such as poor diet and lack of exercise (Renaud and Lorgeril 1992). CVD and the inflammation associated with it often co-presents with other illnesses and can complicate many treatments (Meeus et al. 2000; Poirier et al. 2006). Finding a way to mitigate inflammation is a sought-after way in the prevention and treatment of CVD.
Before exploring potential ways to reduce inflammation, it is important to understand how inflammation impacts the body. Much of the inflammation present in CVDs is linked to some type of damage or injury to the endothelial wall of the heart or an artery (Thom et al. 2006; Greaves and Gordon 2001). One of the most common diseases is valvular heart disease (Greaves and Gordon 2001; Thom et al. 2006). Damaged heart valves can lead to a multitude of problems, such as endocarditis or stenotic valves (Leng et al. 1996). These diseases all involve inflammation of the surrounding tissue, which is one of the hallmarks of the wound healing process (Bao et al. 2009). The acute inflammatory phase of the wound healing process includes blood clotting and the movement of inflammatory cells to the site of the wound, followed by a proliferative phase and a remodeling stage (Christian et al. 2006). The initial inflammatory stage is vital for the later stages to occur and enable wound healing progression (Christian et al. 2006). Angiogenesis, the formation of new blood vessels, is also a key element of wound healing (Nör et al. 1999). Because of the profound widespread effects of inflammation on the body and the body’s innate ability to repair itself through wound healing, many treatments for CVD are negatively impacted by the inflammatory process.
The treatments available for CVD are impacted by inflammation, similarly to how wound healing is. Several treatments for CVD utilize implantable medical devices (biomaterials) which include vascular stents, pacemakers and artificial valves and have been shown to elicit an inflammatory response. The inflammatory process that occurs in the body is often elicited in response to something the body does not recognize, such as these medical devices. These vital treatment strategies result in the host responding to the foreign material, where blood proteins and cells recognize the material as foreign and initiate an immune response. This can lead to a pro-inflammatory state, device failure, loss of therapeutic benefit and ultimately the need for replacement (Slee et al. 2014a). Therefore, an easily accessible strategy that could mitigate inflammation in CVD and lessen the host response to foreign materials would be of significant value to the medical community.
Due to the ease of access from being naturally derived, natural products have become increasingly popular as medicinal options (Jain et al. 2018; Nasri et al. 2014). Many of these products have little scientific data to support much of their purported benefits and they lack US Food and Drug Administration oversight and regulation (Chen and Alpert 2016; Schmitt and Ferro 2013). These compounds may offer an untapped resource for treatments involving the inflammatory process, especially in respect to cell adherence to biomaterials. While the anti-inflammatory effects of some natural compounds have been lightly investigated in previous studies (Sá Coutinho et al. 2018), more evidence is needed on dosage, toxicity and assay variation to confirm proposed use as natural anti-inflammatory drugs (Das and Das 2007). One potential solution can be exemplified through the “French Paradox,” the phenomenon where the French have much lower rates of CVD despite having high fat diets, has been thought to be linked to the daily consumption of red wine (Castaldo et al. 2019; Renaud and Lorgeril 1992). This phenomenon has, in part, been attributed to an antioxidant found in red grapes called resveratrol.
Resveratrol, a polyphenol-derived compound found in the skin of red grapes, is used to make red wine. Studies have shown the anti-inflammatory effects of resveratrol using Tumor Necrosis Factor-α (TNF-α) through the inhibition of intercellular adhesion molecule 1 (ICAM-1), inducible nitric oxide synthase (iNOS) and interleukin-1 beta (IL-1β) messenger RNA (mRNA) expression in human coronary arterial endothelial cells (Huang et al. 2017). Specifically, resveratrol has been shown to improve vascular function by increasing nitric oxide (NO) synthesis and inhibiting its degradation (Förstermann and Li 2011). NO is made and released into endothelial cells by nitric oxide synthase (NOS), which has been found to reduce inflammation and promote vasodilation in the cardiovascular system (Sharma et al. 2007). Along with this, studies have linked NO to immune responses by activated macrophages (Sharma et al. 2007). The large production of NO by activated macrophages allows for their ability to suppress lymphocyte proliferation (Moilanen and Vapaatalo 1995). These qualities that resveratrol has proven to possess are what make it an interesting subject in reducing inflammation.
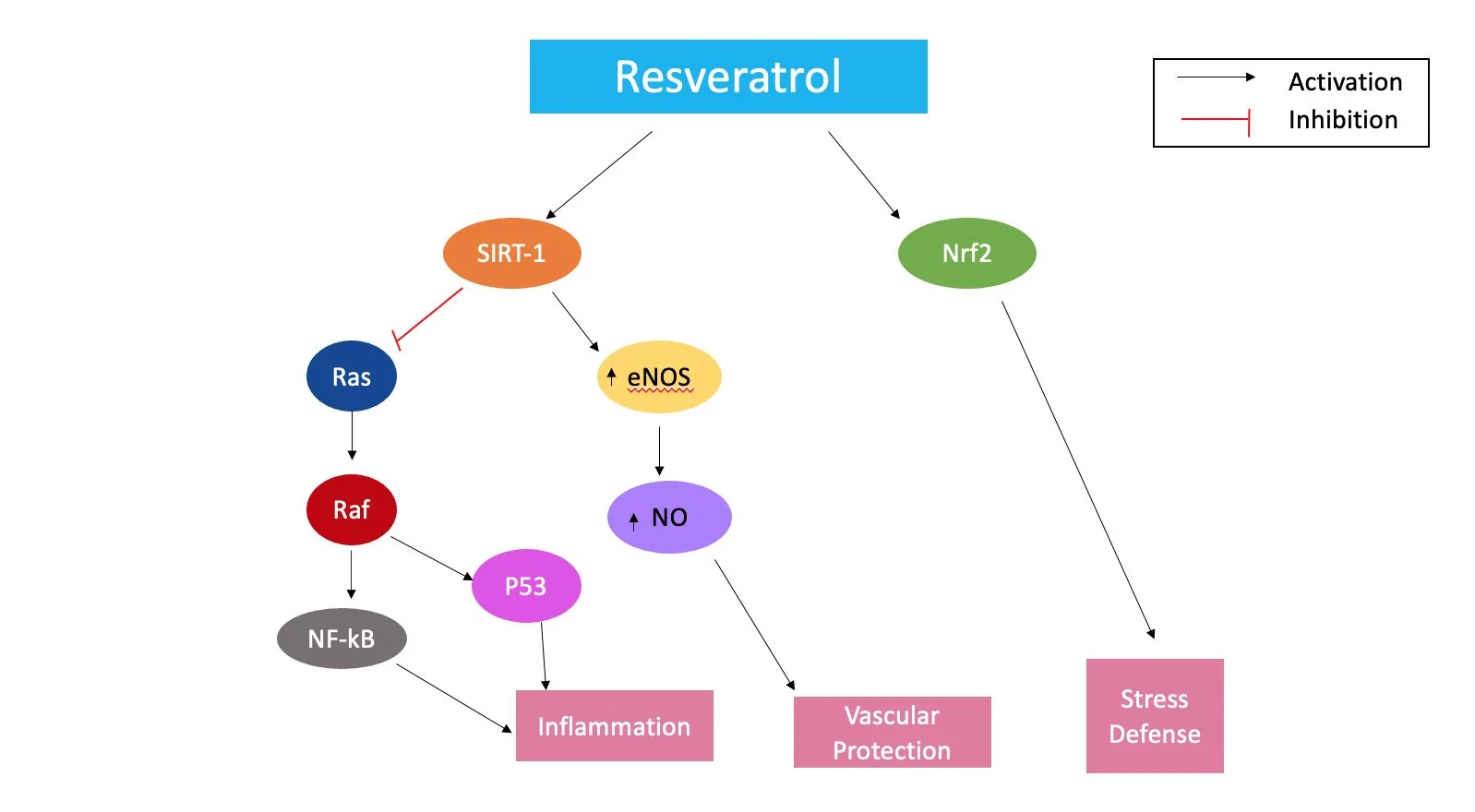
In addition to simply reducing inflammation, the mechanisms by which resveratrol works could be protective in cases of vascular inflammation (Bonnefont-Rousselot 2016; Renaud and de Lorgeril 1992). Resveratrol has been shown to activate several targets, including SIRT-1 and Nrf2, which can upregulate endothelial NOS (eNOS). This, along with other proposed mechanisms of resveratrol’s effects, is demonstrated in Figure 1 (Meng et al. 2021). In addition, several studies have suggested possible cardioprotective effects of resveratrol, including improved endothelial function (Fujitaka et al. 2011) and reduced low-density lipoprotein-cholesterol (LDL-C) (Magyar et al. 2012). Recent reports have suggested that resveratrol enhances the activation of vascular endothelial growth factor expression (VEGF), which stimulates the formation of blood vessels, in turn promoting wound healing, a vital function in the cardiovascular system (Nör et al. 1999). Due to the prevalence of these diseases across the world and the ever-growing need for treatment, it is important for a therapeutic to be readily accessible.
Figure 1. A proposed schematic on the mechanism of Resveratrol’s suppression of the inflammatory response.
Resveratrol has the potential to be a readily accessible treatment for inflammatory diseases in the body since it is a chemical produced in more than 70 plant species in response to environmental stress, such as mechanical injury, microbial infection and UV irradiation (Soleas et al. 1997). Thus, it suggests resveratrol should be studied further because of its possible health implications and the heightened interest in nutraceuticals, or any food substance that contains health benefits, as viable supplements to pharmaceuticals (Jain et al. 2018; Nasri et al. 2014).
To further explore the gaps in the current research on resveratrol, we aimed to use a model for inflammation to test the resveratrol. An established way to model inflammation in vascular endothelial cells is through the use of TNF-α, a common inflammatory cytokine. TNF-α has been shown to greatly increase actin stress fiber accumulation in bovine aortic endothelial cells (BAOECs), mimicking the inflammatory response in the human body (Farwell et al. 2016; Feuerstein et al. 1994). Previous studies have investigated the anti-inflammatory effects of drugs on TNF-α-treated BAOECs, showing a decrease in actin stress fiber accumulation (Farwell et al. 2016; Feuerstein et al. 1994). Resveratrol may possess similar anti-inflammatory qualities when examined in this way. Studies have also shown that implantable materials trigger the host inflammatory response in reaction to the foreign material (Slee et al. 2014a). Macrophages begin to adhere to the implanted material and degrade it, hindering its vital functions (Slee et al. 2014a). Upon treatment with phorbolmyristate acetate (PMA), THP-1 cells, derived from human monocytic leukemia, exhibit a macrophage-like phenotype (Schwager et al. 2017). THP-1 cells are an ideal model for immune responses to implanted biomaterials for this reason (Schwager et al. 2017). Increasing the biocompatibility of implantable materials will lessen the host’s inflammatory response and lead to a longer lifespan of the material.
In order to decrease cell attachment to biomaterials, the Chandler Loop was used as a more physiologically relevant model for the THP-1 cells. The Chandler Loop has shown to be a trusted model for mimicking blood flow over polymers. This mechanism mimics perfusion of blood through medical tubing or blood vessels, making it more physiologically relevant than assays in a petri dish. The tubing or films put inside the tubing, can then be imaged for cell attachment (Slee et al. 2014b).
The conditions in which resveratrol’s effects were explored included endothelial cell stress, endothelial cell injury and biocompatibility. Compared to previous research, this work proposes a novel idea of how beneficial resveratrol could extend the lifespan of biomaterials and reduce rejection of implantable biomaterials through the use of the Chandler Loop assay. The aim of the study is to show that resveratrol displays promising anti-inflammatory effects in these models by reducing the effects of TNF-α on the actin cytoskeleton, decreasing wound healing time and decreasing cell attachment to biomaterials. This will back current research showing the anti-inflammatory effects of resveratrol and expand on it by avoiding negative outcomes of other current treatments, like cell adherence to biomaterials.
Materials and Methods
Cell Culture
Bovine Aortic Endothelial Cells (BAOECs – Cell Applications, San Diego, CA, USA) were cultured using MEM Basal Media (Sigma-Aldrich, St. Louis, MO, USA), 10% heat-inactivated FBS (VWR), 1X Penicillin/Streptomycin (Sigma-Aldrich), 4mM L-Glutamine (Sigma-Aldrich), 1mM sodium pyruvate (Sigma-Aldrich) and 1X MEM nonessential amino acids (Sigma-Aldrich). THP-1 cells (ATCC, Manassas, VA, USA) were cultured using RPMI-1640 (Avantor-VWR, Radnor, PA, USA), 5% FBS (Avantor-VWR), 1X Penicillin/Streptomycin (Sigma-Aldrich) and 0.05mM BME (Sigma-Aldrich). THP-1 cells were differentiated with 160 nM PMA (Sigma-Aldrich) for THP-1 adhesion assays. Cells were grown at 37°C and 5% CO2.
Resveratrol and Endothelial Cell Morphology
BAOECs were seeded on 0.2% bovine gelatin treated coverslips. BAOECs were treated with either 1μM, 10μM or 100μM resveratrol solubilized in 100% ethanol (Spectrum, New Brunswick, NJ, USA) and ethanol-treated BAOECs served as the control were incubated at 37°C for 24 hours. Media were removed and 10mM Cell Tracker Green (Thermo Fisher, Waltham, MA, USA) was added and incubated at 37°C and 5% CO2 for one hour prior to imaging. Cells were fixed in 4% formaldehyde (Thermo Fisher) in 1% BSA/PBS for 10 minutes. Coverslips were mounted using Vectashield plus DAPI (Vector Labs, Burlingame, CA, USA) and imaged using appropriate filters for DAPI and FITC magnification under 400X magnification.
Resveratrol and Cell Stress
BAOECs were seeded on 0.2% bovine gelatin treated coverslips. After 24 hours, two experimental coverslips were treated with 1μM resveratrol (Spectrum) for two hours prior to 0.025μg/mL TNF-α (ProSci, Poway, CA, USA) treatment for 40 minutes. Two TNF-α controls were treated with 0.025μg/mL TNF-α for 40 minutes. One resveratrol control was treated with 1μM resveratrol for two hours. One ethanol-treated coverslip served as the control. Cells were fixed at room temperature in 4% formaldehyde in 1% BSA/PBS with gentle shaking for 10 minutes. Cells were permeabilized with 0.3% Triton-X-100 in 1% BSA/PBS at room temperature with gentle shaking for five minutes. Cells were stained with Alexafluor 488 Phalloidin (Abnova, Taipei, Taiwan) and incubated in the dark at 37°C for 30 minutes. Coverslips were mounted using Vectashield plus DAPI (Vector Labs) and imaged under 400X magnification using appropriate filters for Alexafluor 488 and DAPI. Nuclei were DAPI-counterstained to verify cells but removed post-hoc to allow for black and white image production.
Resveratrol and Wound Healing
BAOECs were seeded on a 0.2% bovine gelatin-treated 6-well plate. Cells were grown to confluency before being incubated in serum-free media for 24 hours. Cells were scraped from a marked area and were treated with either μM, 10μM or 100μM resveratrol (Spectrum) in serum-free media. Ethanol-treated cells served as the control. Cells were incubated at 37°C, 5% CO2 and imaged using phase contrast daily at 100X.
THP-1 Adhesion Assay
The polyurethane films (1 cm X 0.5 cm) used were Tecothane TT1074A (Thermedics, Waltham, MA, USA), a polyether polyurethane. The films were placed in wells of a 24-well TC dish. Three hundred thousand THP-1 cells were activated with 160 nM PMA and added to each well (Stachelek et al. 2005; 2006). Cells were treated with either 1μM, 10μM or 100μM resveratrol (Spectrum) or ethanol as a control. Cells were incubated for three days at 37°C. Cells were fixed at room temperature in 4% formaldehyde in 1% BSA/PBS for 10 minutes. Cells were incubated in Vectashield plus DAPI (Vector Labs) in the dark for 30 minutes at room temperature.
Polyurethane films were imaged under 200X magnification using appropriate filters for DAPI.
Chandler Loop Assay
The Chandler Loop apparatus was assembled as described in Slee et al. (2014b). Briefly, four PVC tubes (Saint Gobain) were filled with 13mL Na-Citrate-treated whole bovine blood (Lampire Biologicals) along with a polyurethane film for easier counting. Cell solutions were treated with either 1μM, 10μM or 100μM resveratrol (Spectrum). One solution was treated with ethanol as a control. The Chandler Loop was run for three hours. Films and tubing sections were fixed at 4°C in 4% formaldehyde in 1% BSA/PBS for 48 hours. Samples of tubing and films were imaged under 200X magnification using filters for DAPI.
Statistics
All statistical significance was determined by a one-way ANOVA with a Tukey post-hoc analysis through Astatsa 2016 Navendu Vasavada. The ANOVA test allows for the determination of differences between the means of multiple treatment groups, while the Tukey post-hoc test controls for Type I errors and helps identify which treatment groups are statistically significant.
Results
Resveratrol does not Significantly Affect Growth or Morphology of BAOECs
BAOECs treated with various resveratrol concentrations showed limited morphological or confluency differences compared to ethanol-treated control cells (Figure 2). These data indicate that the tested concentrations of resveratrol do not significantly impact cell growth or morphology.
Figure 2. The effect of resveratrol on BAOEC morphology. (A) BAOECs cultured in media containing ethanol without resveratrol (B) BAOECs cultured in media containing 1μM resveratrol (C) BAOECs cultured in media containing 10μM resveratrol (D) BAOECs cultured in media containing 100μM resveratrol. Cells were labeled with 10mM Cell Tracker Green. All images were obtained under 400X total magnification. Data representative of three trials.
Resveratrol Decreases Cell Stress in TNF-α-treated BAOECs
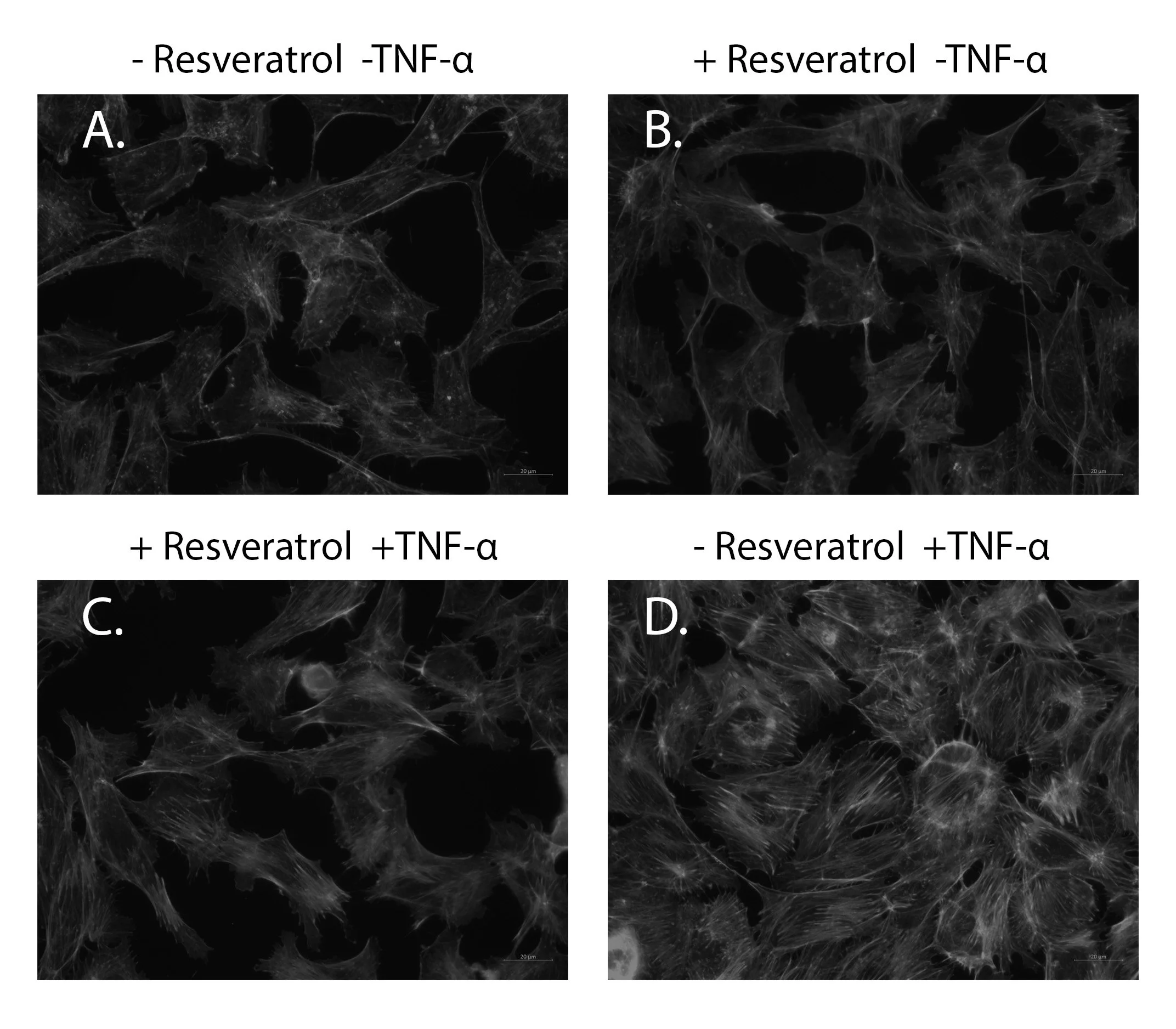
Cell stress has been used as a model for inflammation. TNF-α has been used to stress cells and mimic the effects of inflammation in BAOECs (Farwell et al. 2016). The control cells exhibit little to no actin stress fiber accumulation (Figure 3A). Cells treated with 1μM resveratrol for two hours and no TNF-α show actin stress fiber levels very similar to those of the untreated control, indicating no effect of resveratrol treatment alone (Figure 3B). Cells that were treated with 1μM resveratrol for two hours prior to 0.025μg/mL TNF-α for 40 minutes showed clearly lower levels of actin stress fiber accumulation (Figure 3C) when compared to TNF-α-treated cells (Figure 3D) and a level of stress fibers comparable to the untreated control cells (Figure 3A). When cells were exposed to 0.025μg/mL TNF-α for 40 minutes and no resveratrol, a noticeable increase in actin stress fiber accumulation is apparent (Figure 3D).
Figure 3. The effect of resveratrol on cell stress. BAOECs were cultured on 0.2% bovine gelatin coated glass coverslips and were (A) ethanol- treated control, (B) treated with 1μM resveratrol for two hours, (D) 0.025μg/mL TNF-α for 40 min, or (C) 1μM resveratrol for two hours prior to 0.025μg/mL TNF-α for 40 min. All images were obtained using DAPI and Alexafluor 488 filters under 400X total magnification. Image analysis settings were set for the Resveratrol-TNFα control (A) and remained the same throughout each replicate. Data representative of three trails. DAPI counterstain used to verify cell nuclei was removed to allow for black and white image preparation. For image clarity, all images were modified (midtones: 1.04, highlights 255, and saturation -100).
Resveratrol decreases wound healing time in vitro for BAOECs
By Day 4, investigation of in vitro wound healing revealed all three concentrations of resveratrol-treated cells had completely healed the wound, while the untreated control did not (Figure 4). The effects of resveratrol could be seen as early as Day 2, where cells could be seen migrating into the wounded area, whereas virtually none of the untreated control cells migrated into the area.
Figure 4. The effect of resveratrol on wound healing. BAOECs were grown in 0.2% bovine gelatin-treated 6 well tissue culture dishes to confluency. At confluency, the cells were serum-starved for 24 hours, followed by wounding with a cell scrapper. Phase contrast images were taken every 24 hours to track the migration into the wounded area. Representative images taken under 100X of 10 replicates. For clarity, all images were enhanced (brightness 109, contrast 44).
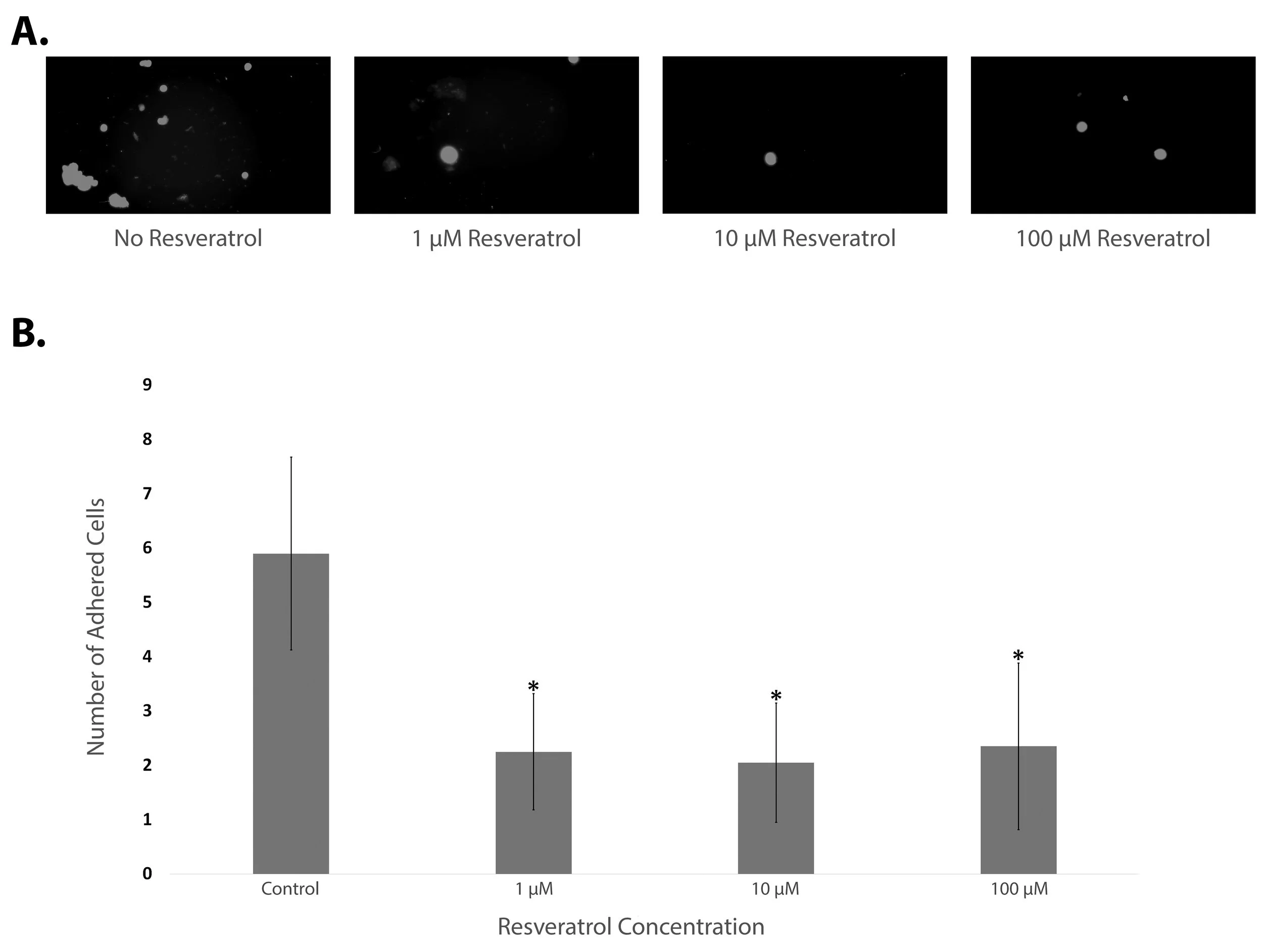
Resveratrol Limits THP-1 Adhesion to a Biomaterials
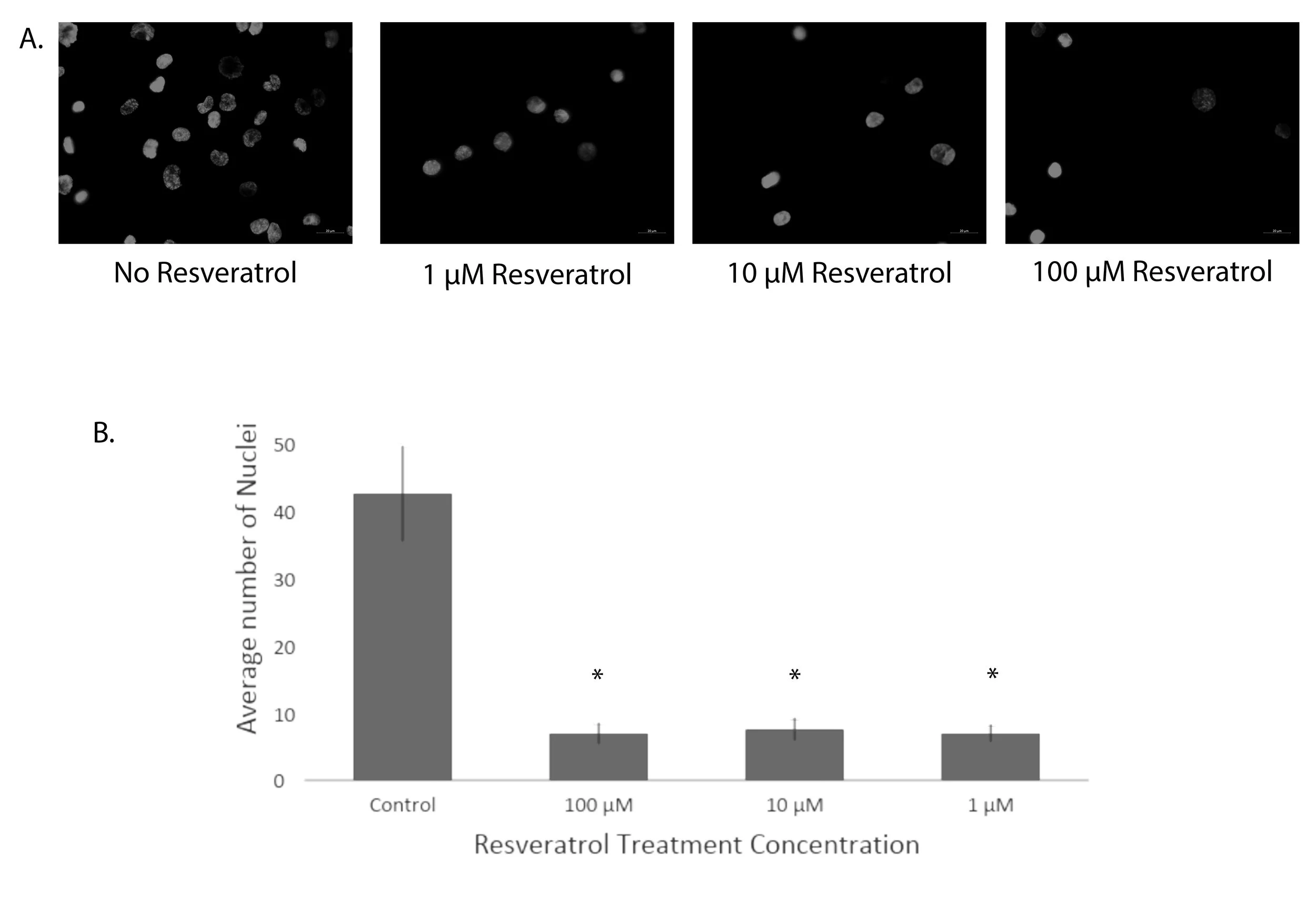
PMA-activated THP-1 cells exhibit a macrophage-like morphology and will adhere to foreign materials like polyurethane. Randomly imaged areas of polyurethane under 200X magnification allow for quantification of the number of adhered cells to the polyurethane (Figure 5A). Average manual cell counts are shown (Figure 5B). All three concentrations of resveratrol result in statistically significant (p < 0.01) decreases in THP-1 cell adhesion to polyurethane compared to control, decreasing the average number of cells from 40 cells to less than 10 cells.
Figure 5. The effect of resveratrol on THP-1 cell adhesion. THP-1 cells were incubated with the indicated concentration of resveratrol during the assay compared to an ethanol-treated control and imaged under 200X magnification with DAPI filters. (A) Representative images of DAPI-stained nuclei of adhered THP-1 cells to polyurethane films. (B) Graphical representation of cell counts from 20 replicates, showing mean ± standard deviation. * Statistical significance determined by one-way ANOVA with a Tukey post-hoc analysis (p < 0.01).
Resveratrol Continues to Exhibit Cell Adhesion Protection in Physiologically Relevant Model
A more physiologically relevant model of biomaterial rejection involves the application of a Chandler Loop Apparatus to move blood over the polyurethane films as it would in vivo (Slee et al. 2014b). Average cell counts on the polyurethane films are shown (Figure 6B). All three concentrations of resveratrol result in statistically significant (p < 0.01) decreases in cell adhesion to polyurethane compared to control, decreasing cell attachment from an average of six cells in the control to an average of two cells in the treated films.
Figure 6. The effect of resveratrol on whole bovine blood cell adhesion in a Chandler Loop apparatus. Whole bovine blood was perfused through PVC tubing containing polyurethane films with the indicated concentration of resveratrol during the assay compared to ethanol-treated control imaged under 200X magnification with DAPI filters. (A) Representative images of DAPI-stained nuclei of adhered THP-1 cells to polyurethane films after Chandler Loop exposure. (B) Graphical representation of cell counts from 20 replicates, showing mean ± standard deviation. * Statistical significance relative to the control determined by one-way ANOVA with a Tukey post-hoc analysis (p < 0.01).
Discussion
In this study, we sought to investigate resveratrol as a possible anti-inflammatory that could have beneficial effects in the treatment of CVD, but also attenuate the host response to foreign materials, which can complicate the co-presenting diseases. An established model of cell stress using TNF-α (Farwell et al. 2016) demonstrated that resveratrol significantly attenuated cell stress in sub-confluent BAOECs. Confirming previous research, resveratrol visibly reduced the amount of actin stress fiber production by TNF-α, indicating that it possesses anti-inflammatory characteristics. This further supports earlier findings showing resveratrol’s anti-inflammatory properties and ability to modulate inflammatory pathways (Sá Coutinho et al. 2018; Huang et al. 2017). Thus, resveratrol shows promise as an anti-inflammatory drug, particularly in the area of wound healing.
While the modulation of inflammatory pathways is of high interest in terms of resveratrol’s properties, resveratrol has also begun to be studied for its wound healing properties. It has been shown that resveratrol can play a factor in wound healing by preventing scar formation (Zhao et al. 2020). While resveratrol’s effects on the promotion of wound healing have been demonstrated, we examined the impacts of resveratrol with respect to dosing and time taken for a wound to heal. Using a scratch wound healing assay, it was shown that resveratrol significantly improved wound healing compared to control in a much quicker period of time, suggesting that it prevents inflammation while still promoting the wound healing process. Reducing the amount of time it takes for a wound to heal implies a quicker reduction in inflammation of the wound and overall quicker healing times, which is advantageous in the case of CVD among other diseases.
Resveratrol has not only been shown to be anti-inflammatory and aid in the wound healing process, but through our work, we have shown that resveratrol treatment of confluent BAOECs does not harm normal cell morphology. This finding is important because it shows that resveratrol will not have any adverse effects on healthy cell morphology. This confirms previous research showing that resveratrol has little to no effect on normal cell morphology (Salehi et al. 2018). This is particularly crucial when it comes to managing biomaterial rejection.
In the case of biomaterial rejection, inflammation can be damaging to the material and the body if left uncontrolled (Browne and Pandit 2015). Although research has shown different mechanisms for reducing cell attachment to biomaterials, such as polymers, there is limited research available on resveratrol’s ability to attenuate cell attachment to biomaterials (Stachelek et al. 2011). The results shown contribute a novel finding to show resveratrol’s promise in attenuating cell attachment to biomaterials. As previously mentioned, the Chandler Loop has been shown to be a trusted model for mimicking blood flow over polymers. Through THP-1 cell adhesion assays and Chandler Loop assays with whole bovine blood, it was shown that resveratrol significantly decreased the number of adhered cells to polyurethane, a common polymer used in clinical settings, suggesting that resveratrol prevents inflammatory cells from adhering to polymeric materials. Taken together, the results suggest that passive addition of resveratrol to blood or cell culture medium has a significant in vitro therapeutic effect by being a possible way to extend the lifespan of implantable biomaterials by reducing inflammatory cell attachment to the biomaterials.
Conclusion
A main limitation of this study was the lack of in vivo experiments. In addition, experiments with endothelial cells were performed using bovine cell lines, as opposed to human cells. Future research should focus on supporting this three pronged approach in an in vivo model system, through the use of human cell lines and animal models. Using human and animal models is a more in-depth approach than our in vitro models, but would allow this compound to be closer to therapeutic use. In the long term implications of taking this research further, expanding the lifespan of implantable biomaterials would pose a benefit to the patient in the reduction of procedures needed, as well as an overall reduction in the cost of replacing implantable biomaterials more frequently. However, the data presented herein suggest that resveratrol may ameliorate potentially damaging inflammation related to CVD and biomaterial rejection.
In conclusion, resveratrol shows promise as a possible therapeutic agent for vascular inflammation, as well as biomaterial rejection in vitro. This work helps to aid in the confirmation of previous studies to solidify resveratrol as an anti-inflammatory agent by testing different concentrations of the chemical. In addition, it reveals the potential impact of resveratrol on several functions of endothelial cells, including wound healing and response to biomaterials, not previously shown. These results were consistent and expected based on the anti-inflammatory properties shown by resveratrol in previous research. While reducing inflammation is favorable for many diseases, resveratrol’s ability to reduce inflammation, reduce the time needed for complete wound healing and prevent inflammatory cell attachment to implantable biomaterials makes it an interesting area of study for biomaterials and organ transplantation.
Conflict of Interests
The authors declare that there are no conflicts of interest.
Acknowledgements
The authors thank Allison Mattern and Carmen Azar for critical manuscript review. The authors wish to thank Dr. Stanley J. Stachelek of the Children’s Hospital of Philadelphia for the polyurethane films used in the THP-1 adhesion assays and Dr. Linda Lowe-Krentz of Lehigh
University for the BAOECs. This research was financially supported by DeSales University,
DeSales University Department of Biology, the DeSales University Dr. Harry Warren
Endowment for Natural Science Equipment, the Dr. Rodger Berg Endowment for Excellence in Undergraduate Natural Science Education and Women for DeSales, a philanthropic group dedicated to the success of DeSales’ students.
References
Bao, P., Kodra, A., Tomic-Canic, M., Golinko, M. S., Ehrlich, H. P. and Brem, H. (2009) ‘The Role of Vascular Endothelial Growth Factor in Wound Healing’, Journal of Surgical Research, 153(2), pp. 347–358, available: https://doi.org/10.1016/j.jss.2008.04.023.
Bonnefont-Rousselot, D. (2016) ‘Resveratrol and Cardiovascular Diseases’, Nutrients, 8(5), p. E250, available: https://doi.org/10.3390/nu8050250.
Browne, S. and Pandit, A. (2015) ‘Biomaterial-mediated modification of the local inflammatory environment’, Frontiers in Bioengineering and Biotechnology, 3, p. 67, available: https://doi.org/10.3389/fbioe.2015.00067.
Castaldo, L., Narváez, A., Izzo, L., Graziani, G., Gaspari, A., Minno, G. D. and Ritieni, A. (2019) ‘Red Wine Consumption and Cardiovascular Health', Molecules (Basel, Switzerland), 24(19), p. E3626, available: https://doi.org/10.3390/molecules24193626.
Chen, Q.M. and Alpert, J.S. (2016) 'Nutraceuticals: Evidence of Benefit in Clinical Practice?', The American Journal of Medicine, 129(9), pp. 897–898, available: https://doi.org/10.1016/j.amjmed.2016.03.036.
Christian, L.M., Graham, J. E., Padgett, D. A., Glaser, R. and Kiecolt-Glaser, J. K. (2006) 'Stress and Wound Healing', Neuroimmunomodulation, 13(5–6), pp. 337–346, available: https://doi.org/10.1159/000104862.
Das, S. and Das, D.K. (2007) 'Anti-inflammatory responses of resveratrol', Inflammation & Allergy Drug Targets, 6(3), pp. 168–173, available: https://doi.org/10.2174/187152807781696464.
Farwell, S.L.N., Kanyi, D., Hamel, M., Slee, J. B., Miller, E. A., Cipolle, M. D. and Lowe-Krentz, L. J. (2016) 'Heparin Decreases in Tumor Necrosis Factor α (TNFα)-induced
Endothelial Stress Responses Require Transmembrane Protein 184A and Induction of Dual Specificity Phosphatase 1', Journal of Biological Chemistry, 291(10), pp. 5342–5354, available: https://doi.org/10.1074/jbc.M115.681288.
Feuerstein, G.Z., Liu, T. and Barone, F.C. (1994) 'Cytokines, inflammation, and brain injury: role of tumor necrosis factor-alpha', Cerebrovascular and Brain Metabolism Reviews, 6(4), pp. 341–360, available: PMID: 7880718
Förstermann, U. and Li, H. (2011) 'Therapeutic effect of enhancing endothelial nitric oxide synthase (eNOS) expression and preventing eNOS uncoupling: Prevention of eNOS uncoupling', British Journal of Pharmacology, 164(2), pp. 213–223, available: https://doi.org/10.1111/j.1476-5381.2010.01196.x.
Fujitaka, K., Otani, H., Jo, F., Jo, H., Nomura, E., Iwasaki, M., Nishikawa, M., Iwasaka, T. and Das, D. K. (2011) 'Modified resveratrol Longevinex improves endothelial function in adults with metabolic syndrome receiving standard treatment', Nutrition Research (New York, N.Y.), 31(11), pp. 842–847, available: https://doi.org/10.1016/j.nutres.2011.09.028.
Greaves, D.R. and Gordon, S. (2001) 'Immunity, atherosclerosis and cardiovascular disease', Trends in Immunology, 22(4), pp. 180–181, available: https://doi.org/10.1016/S14714906(00)01848-2.
Huang, F.-C., Kuo, H. C., Huang, Y. H., Yu, H. R., Li, S. C., and Kuo, H. C. (2017) 'Anti-inflammatory effect of resveratrol in human coronary arterial endothelial cells via induction of autophagy: implication for the treatment of Kawasaki disease', BMC pharmacology & toxicology, 18(1), p. 3, available: https://doi.org/10.1186/s40360-016-0109-2
Jain, S., Buttar H.S., Chintameneni, M. and Kaur, G. (2018) 'Prevention of Cardiovascular Diseases with Anti-Inflammatory and Anti- Oxidant Nutraceuticals and Herbal Products: An Overview of Pre-Clinical and Clinical Studies', Recent Patents on Inflammation & Allergy Drug Discovery, 12(2), pp. 145–157, available: https://doi.org/10.2174/1872213X12666180815144803.
Leng, G.C., Lee, A. J., Fowkes, F. G., Whiteman, M., Dunbar, J., Housley, E., and Ruckley, C. V. (1996) 'Incidence, Natural History and Cardiovascular Events in Symptomatic and Asymptomatic Peripheral Arterial Disease in the General Population', International Journal of Epidemiology, 25(6), pp. 1172–1181, available: https://doi.org/10.1093/ije/25.6.1172.
Magyar, K., Halmosi, R., Palfi, A., Feher, G., Czopf, L., Fulop, A., Battyany, I., Sumegi, B., Toth, K., and Szabados, E. (2012) 'Cardioprotection by resveratrol: A human clinical trial in patients with stable coronary artery disease', Clinical Hemorheology and Microcirculation, 50(3), pp. 179– 187, available: https://doi.org/10.3233/CH-2011-1424.
Meeus, F., Kourilsky, O., Guerin, A. P., Gaudry, C., Marchais, S. J., and London, G. M. (2000) 'Pathophysiology of cardiovascular disease in hemodialysis patients', Kidney International. Supplement, 76, pp. S140-147, available: https://doi.org/10.1046/j.1523-1755.2000.07618.x.
Meng, T., Xiao, D., Muhammed, A., Deng, J., Chen, L., and He, J. (2021) 'Anti-Inflammatory Action and Mechanisms of Resveratrol', Molecules (Basel, Switzerland), 26(1), p. E229, available: https://doi.org/10.3390/molecules26010229.
Moilanen, E. and Vapaatalo, H. (1995) 'Nitric Oxide in Inflammation and Immune Response', Annals of Medicine, 27(3), pp. 359–367, available: https://doi.org/10.3109/07853899509002589.
Nasri, H., Baradaran, A., Shirzad, H., and Rafieian-Kopaei, M. (2014) 'New concepts in nutraceuticals as alternative for pharmaceuticals', International Journal of Preventive Medicine, 5(12), pp. 1487–1499, available: PMID: 25709784
Nör, J.E., Christensen, J., Mooney, D. J., and Polverini, P. J. (1999) 'Vascular Endothelial Growth Factor (VEGF)-Mediated Angiogenesis Is Associated with Enhanced Endothelial Cell Survival and Induction of Bcl-2 Expression', The American Journal of Pathology, 154(2), pp. 375–384, available: https://doi.org/10.1016/S0002-9440(10)65284-4.
Poirier, P., Giles, T. D., Bray, G. A., Hong, Y., Stern, J. S., Pi-Sunyer, F. X., Eckel, R. H., American Heart Association, and Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism (2006) 'Obesity and Cardiovascular Disease: Pathophysiology, Evaluation, and Effect of Weight Loss: An Update of the 1997 American Heart Association Scientific Statement on Obesity and Heart Disease from the Obesity Committee of the Council on Nutrition, Physical Activity, and Metabolism', Circulation, 113(6), pp. 898–918, available: https://doi.org/10.1161/CIRCULATIONAHA.106.171016.
Renaud, S. and Lorgeril, M. (1992) 'Wine, alcohol, platelets, and the French paradox for coronary heart disease', The Lancet, 339(8808), pp. 1523–1526, available: https://doi.org/10.1016/0140-6736(92)91277-F.
Sá Coutinho, D., Pacheco, M. T., Frozza, R. L., and Bernardi, A. (2018) 'Anti-Inflammatory Effects of Resveratrol: Mechanistic Insights', International Journal of Molecular Sciences, 19(6), p. E1812, available: https://doi.org/10.3390/ijms19061812.
Salehi, B., Mishra, A. P., Nigam, M., Sener, B., Kilic, M., Sharifi-Rad, M., Fokou, P. V. T., Martins, N., and Sharifi-Rad, J. (2018) 'Resveratrol: A Double-Edged Sword in Health Benefits', Biomedicines, 6(3), p. 91, available: https://doi.org/10.3390/biomedicines6030091.
Schmitt, J. and Ferro, A. (2013) 'Nutraceuticals: is there good science behind the hype?: Editorial', British Journal of Clinical Pharmacology, 75(3), pp. 585–587, available: https://doi.org/10.1111/bcp.12061.
Schwager, J. Richard, N., Widmer, F., and Raederstorff, D. (2017) 'Resveratrol distinctively modulates the inflammatory profiles of immune and endothelial cells', BMC Complementary and Alternative Medicine, 17(1), p. 309, available: https://doi.org/10.1186/s12906-017-1823-z.
Sharma, J.N., Al-Omran, A. and Parvathy, S.S. (2007) 'Role of nitric oxide in inflammatory diseases', Inflammopharmacology, 15(6), pp. 252–259, available: https://doi.org/10.1007/s10787-007-0013-x.
Shiels, M.S., Haque, A. T., Berrington de González, A., and Freedman, N. D. (2022) 'Leading Causes of Death in the US During the COVID-19 Pandemic, March 2020 to October 2021', JAMA Internal Medicine, 182(8), p. 883, available: https://doi.org/10.1001/jamainternmed.2022.2476.
Slee, J.B., Christian, A. J., Levy, R. J., and Stachelek, S. J. (2014a) 'Addressing the Inflammatory Response to Clinically Relevant Polymers by Manipulating the Host Response Using ITIM Domain-Containing Receptors', Polymers, 6(10), pp. 2526–2551, available: https://doi.org/10.3390/polym6102526.
Slee, J.B., Alferiev, I. S., Levy, R. J., and Stachelek, S. J. (2014b) 'The Use of the Ex Vivo Chandler Loop Apparatus to Assess the Biocompatibility of Modified Polymeric Blood Conduits', Journal of Visualized Experiments, (90), p. 51871, available: https://doi.org/10.3791/51871.
Soleas, G.J., Diamandis, E.P. and Goldberg, D.M. (1997) 'Resveratrol: A molecule whose time has come? And gone?', Clinical Biochemistry, 30(2), pp. 91–113, available: https://doi.org/10.1016/S0009-9120(96)00155-5.
Stachelek, S.J., Alferiev, I., Choi, H., Kronsteiner, A., Uttayarat, P., Gooch, K. J., Composto, R. J., Chen, I. W., Hebbel, R. P., and Levy, R. J. (2005) 'Cholesterol-derivatized polyurethane: Characterization and endothelial cell adhesion', Journal of Biomedical Materials Research Part A, 72A(2), pp. 200– 212, available: https://doi.org/10.1002/jbm.a.30224.
Stachelek, S.J., Alferiev, I., Choi, H., Chan, C. W., Zubiate, B., Sacks, M., Composto, R., Chen, I. W., and Levy, R. J. (2006) 'Prevention of oxidative degradation of polyurethane by covalent attachment of di‐ tert ‐butylphenol residues', Journal of Biomedical Materials Research Part A, 78A(4), pp. 653–661, available: https://doi.org/10.1002/jbm.a.30828.
Stachelek, S.J., Finley, M. J., Alferiev, I. S., Wang, F., Tsai, R. K., Eckells, E. C., Tomczyk, N., Connolly, J. M., Discher, D. E., Eckmann, D. M., and Levy, R. J. (2011) 'The effect of CD47 modified polymer surfaces on inflammatory cell attachment and activation', Biomaterials, 32(19), pp. 4317–4326, available: https://doi.org/10.1016/j.biomaterials.2011.02.053.
Thom, T., Haase, N., Rosamond, W., Howard, V. J., Rumsfeld, J., Manolio, T., Zheng, Z. J., Flegal, K., O'Donnell, C., Kittner, S., Lloyd-Jones, D., Goff, D. C., Jr, Hong, Y., Adams, R., Friday, G., Furie, K., Gorelick, P., Kissela, B., Marler, J., Meigs, J., … American Heart Association Statistics Committee and Stroke Statistics Subcommittee(2006) 'Heart disease and stroke statistics--2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee', Circulation vol. 113,6, e85-151, available: doi:10.1161/CIRCULATIONAHA.105.171600
Zhao, C.-C., Zhu, L., Wu, Z., Yang, R., Xu, N. and Liang L. (2020) 'Resveratrol-loaded peptide-hydrogels inhibit scar formation in wound healing through suppressing inflammation', Regenerative Biomaterials, 7(1), pp. 99–107, available: https://doi.org/10.1093/rb/rbz041.