William Gross1, Alexis Voulgaropoulos1, Dilara Sen1, Albert Keung1*
1Department of Chemical & Biomolecular Engineering, North Carolina State University
Abstract
There is an increasing appreciation of the influence of physical factors on neural stem cell biology. 2D experiments have provided compelling evidence that neural stem cell development is directed in part by mechanobiological influences, which refer to the stimuli a cell experiences as a result of movement, geometry or substrate material. As a result, human cerebral organoids are an exciting model to expand on such mechanobiological studies. Their three-dimensional nature makes them suitable for exploring the potential impacts of physical conditions such as spatial geometry and mechanical stress, capturing the complex progression of stem and progenitor cells into neurons and more accurately mimicking in vivo conditions. In this study, we leverage these strengths by using human cerebral organoids to explore how physical constraint influences the morphological development of human brain tissue. Cerebral organoid precursors were seeded into geometrically distinct microwells and allowed to develop until they exceeded the size of their wells, after which they were released from the constraints. These samples were then allowed to freely mature for a maximum time of 40 days post-confinement, after which they were stained with fluorescent antibodies to highlight neural progenitor cell presence and organization. We report that organoids derived from precursors, confined in microwells with four convex points, exhibited deterioration of stem cell structures known as ‘neural rosettes’ at a rate faster than those cultured in round microwells. Our findings suggest that microtissue geometry may inhibit neural progenitor proliferation and/or organization, resulting in smaller rosettes and a loss of rosette maintenance, even after organoids are removed from confinement. Further investigation is needed to dissect and decouple the potential effects geometry has on paracrine morphogen release and diffusion, mechanical stress and strain and the time points where these effects have the greatest biological impact.
Introduction
The influence of biochemical factors on neural development has been a central focus of research for many decades (Zamenhof and Van Marthens 1974; Briscoe and Ericson 1999; Fukuchi-Shimogori and Grove 2001; Georgieff 2007). However, it has since become apparent that physical factors such as stresses, strains and the natural shape of tissues are also important in neural development (Moore and Sheetz 2011; Sokol 2016; Stukel and Willits 2016; Chighizola et al. 2019). In particular, neural stem cell (NSC) differentiation can be biased towards either neuronal or glial fates depending on the stiffness of its substrate (Saha et al. 2008). Similar changes in NSC fate can be caused by the geometric arrangement of the substrate, with patterns that maximize cell-cell contact resulting in an upregulation of neuronal fates and increased rates of NSC proliferation (Solanki et al. 2010; Kijeńska et al. 2012; Chen et al. 2014). The vast majority of this work has been performed in 2D systems, using homogenous cultures of neural stem cells. As a result, the ability to study the effects of these physical factors is often limited to the cellular level. Human cerebral organoids (hCOs), which are 3D tissue cultures containing microstructures analogous to those in the developing human brain, provide an opportunity for researchers to explore these mechanobiological factors in a far more complex environment. hCOs exhibit tissue-level morphology, and their 3-dimensional nature allows them to model mechanical effects in all of the dimensions present during in vivo development.
Our research group previously investigated the transcriptomic effects of geometrically confining organoid precursors known as embryoid bodies (EBs) (Sen, Voulgaropoulos and Keung 2021). During these experiments, stem cells were differentiated into EBs within agarose microwells of different geometric shapes but identical volumes for 5 days post-seeding. The EBs were subsequently removed from the wells and differentiated into hCOs for 10, 20, 30 or 40 days, inclusive of the five days in the microwells. The transcriptomic profiles of these hCOs were assessed by RNAseq, revealing differences in gene expression between geometric conditions, which correlated with potentially altered cellular compositions within these hCOs. The greatest differences in expression were observed between organoids seeded in round wells, which were used as a control, and those from ‘butterfly-shaped’ wells featuring four points of geometric deformation. The expression of mechanotransduction-associated genes was significantly upregulated in butterfly-well organoids.
Based upon the observations of changes in transcriptomic signatures associated with cell type compositions, we hypothesized that changes in cellular composition may be reflected and easily observed through morphological and microstructural changes in hCOs. Because Sen and colleagues (2021) observed the greatest transcriptomic differences between organoids seeded in round wells and those in butterfly-shaped wells, we immunohistochemically stained hCOs from these two experimental groups. After investigating several different characteristics and cell types, we found that the most prominent phenotypical contrast appeared in the expression of ring-shaped regions of SOX2+ neural progenitor cells known as neural rosettes. These features are believed to be analogous to the neural tube, a structure that appears during in vivo embryonic development and eventually forms into the brain and spinal cord (Zhang et al. 2001). Butterfly hCOs exhibited both smaller rosettes and a greater quantity of unstructured SOX2 aggregates than their round counterparts, showing that geometric confinement alters hCO development, supporting the hypothesis that mechanobiological effects are a relevant factor in neural development. Our results expand our understanding of this process, highlighting the promise of geometric factors as a tool for engineering cerebral tissue and as a new lens through which to study neural-tube-related diseases.
Results
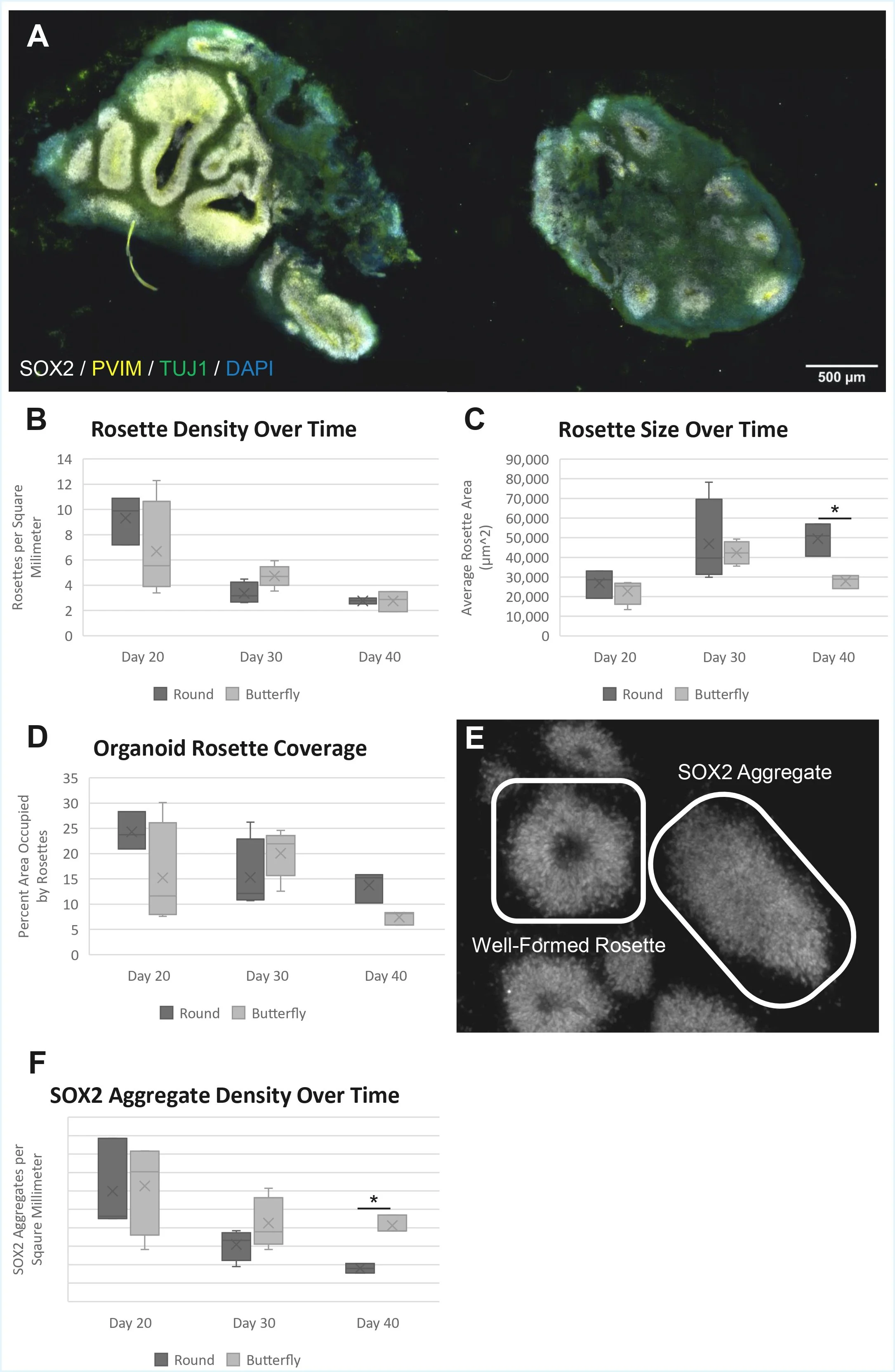
hCOs generated from round microwells maintain rosette structures and growth longer than those generated from butterfly hCOs. The present work utilized fixed and frozen samples generated from our prior study (Sen et al. 2021). Day 10, 20, 30 and 40 hCOs derived from embryoid bodies confined in butterfly and round microwells were cryosectioned and immunostained for radial glia neural stem cells (phospho-vimentin), neural progenitors (SOX2), neurons (TUJ1) and cell nuclei (DAPI). By day 20, all hCOs had developed neural rosettes, with round-well and butterfly hCOs containing similar numbers of rosettes normalized by area (Figure 1B; t-test, df = 5, p = .295, two-tailed). Between days 20 and 40, the number of rosettes decreased in both groups (Figure 1B). However, while the average rosette size increased in both hCO groups up to day 20, the two sets diverged from this point, with round hCOs maintaining their rosette sizes and butterfly hCO rosettes becoming smaller in size at day 40 (Figure 1C; t-test, df = 4, p = .0316, two-tailed).
Although the number of rosettes decreased in both round and butterfly hCOs, the fraction of organoid area occupied by SOX2+ rosettes in round-well hCOs remained approximately equal between days 30 and 40, due to the loss in rosette numbers being counteracted by the increased size of the round-well rosettes (Figure 1D). This suggests that the rosettes in round hCOs merged as they grew. This is further supported by the irregular shapes of rosettes in this group, which may have developed as smaller rosettes coalesced. Meanwhile, in butterfly hCOs, the absolute area occupied by rosettes shrank over time alongside the number of rosettes.
We also noticed circular patches of SOX2+ neural stem cells without lumens. These are most likely rosette precursors or former rosettes that had begun to lose their structure. We decided to refer to these features as “SOX2 aggregates” and recorded them separately from well formed rosettes (Figure 1E). Twenty days post-confinement, both round and butterfly hCOs expressed a similar number of SOX2 aggregates. However, by day 40, the butterfly hCOs contained over twice the number of aggregates (Figure 1F; Mann-Whitney, U = 0, n1 = n2 = 3, p = .0495), supporting the conclusion that these hCOs are less capable of driving and maintaining growth of rosette structures.
Figure 1. (A) Organoids 40 days post-confinement in (left) round and (right) butterfly wells. Samples stained with antibodies to highlight (sex-determining region Y)-box 2 (SOX2), white; phosphorylated vimentin (pVIM), yellow; and class III β-tubulin (TUJ1), green. 4′,6-diamidino-2-phenylindole (DAPI) used to stain DNA-rich regions, represented in blue. (B) Comparison of rosette density in round-and butterfly-well organoids. n = 3-5 organoids per point. Two-tailed t-test results: Day 20: df = 5, p = .295; Day 30: df =7, p = .0504; Day 40: df = 4, p = .994. (C) Size comparison of rosettes in round- and butterfly-well organoids. n = 3-5 organoids per point. Two-tailed t-test results: Day 20: df = 5, p = .298; Day 30: df = 7, p = .306 Day 40: df = 4, p = .0316. (D) Comparison of relative area occupied by SOX2+ rosettes in round- and butterfly-well organoids. n = 3 to 5 organoids per point. Two-tailed t-test results: Day 20: df = 5, p = .620; Day 30: df = 7, p = .310; Day 40: df = 4, p = .0529. (E) Examples of a well-formed rosette and a SOX2 aggregate. (F) Comparison of densities of SOX2 aggregates. n = 3-5 organoids per point Mann-Whitney test results: Day 20: U = 6, n1 = 3, n2 = 4, p = 1; Day 30: U = 6, n1 = 4, n2 = 5, p = .327; Day 40: U = 0, n1 = n2 = 3, p = .0495. *Two-tailed t-test or Mann-Whitney test with p<0.05.
Discussion
These results show that early geometric confinement of EBs has a noticeable effect on neural rosette morphology and that these effects take time to visibly develop. At day 20, no significant differences were found between the two groups, but, by day 40, two distinct phenotypes were evident. Round hCOs had larger rosettes with defined lumens, while the rosettes of butterfly hCOs decreased in size and structural definition. This resulted in a greater number of SOX2 aggregates and overall fewer rosettes.
There are two natural processes through which neural rosette size might change. The first is through the division of neuroepithelial cells (Elkabetz et al. 2008). When these neural progenitors divide symmetrically, they produce two multipotent daughter cells, causing the rosette to increase in size. Conversely, asymmetric division of neuroepithelial cells causes rosettes to stabilize or shrink. During these types of divisions, one progenitor cell creates one differentiated daughter and one progenitor cell, or two differentiated daughter cells, with daughter cells differentiating and migrating away from the lumen (Elkabetz et al. 2008; Banda et al. 2015). Consistent with our observations, Sen and colleagues found that butterfly hCOs display an upregulation in differentiation-related genes at day 10, while round hCOs experience the same level of upregulation at day 20 (Sen et al. 2021). This suggests that downstream effects of early geometric confinement can accelerate the differentiation timeline and may explain the disappearance of rosettes in butterfly hCOs. Future experiments with longer post confinement periods would be necessary to determine if round hCO rosettes are bound to the same fate as their butterfly counterparts, though on a delayed timescale, or if they will continue to maintain their structure. The second mechanism that may contribute to observed differences in rosette size is cell apoptosis. Future experiments could track caspase-3 levels to determine the extent to which apoptosis is affected by early geometric confinement.
The effects of early confinement on these processes is also a topic of interest. One possible explanation is that the interior curves of the butterfly-shaped microwell exert pressure on the developing embryoid body, triggering mechanotransductive signaling within the cells. Mechanosensitive Notch and Rho/ROCK signaling pathways have both been implicated in rosette maintenance (Elkabetz et al. 2008; Medelnik et al. 2018; Townshend et al. 2020; Main et al. 2013; Loerakker et al. 2018; Totaro et al. 2017). While Sen and colleagues (2021) found no significant differences in the transcriptomic expression of mechanotransductive genes, regulation through these signaling pathways could occur largely at the protein and post translational level. In addition, Sen and colleagues (2021) did reveal a considerable upregulation in Wnt signaling in butterfly hCOs, which is also associated with mechanotransductive sensing and signaling (Cha and Srinivasan 2016; Cha et al. 2016). This pathway is known to relate to rosette morphology in several ways. Firstly, the inhibition of Wnt results in larger rosette sizes (Elkabetz et al. 2008). Secondly, this pathway upregulates the GLI3 gene, which represses expression of the morphogen Sonic Hedgehog (SHH) (Li et al. 2009). SHH has been known to increase lumen size by inducing proliferation (Elktabetz et al. 2008). Collectively, this prior work suggests that the mechanosensitive upregulation of Wnt may reduce stem cell proliferation in butterfly-well organoids, leading to the observed reduction in rosette structure and size. This hypothesis could be tested with experiments manipulating the Wnt pathway in round-well, butterfly-well and non-confined rosettes. For instance, employing Wnt pathway inhibitors such as WIF-1 and agonists such as GSK-3β inhibitors could demonstrate whether Wnt upregulation produces rosette phenotypes similar to those found in butterfly-well organoids (Cruciat and Niehrs 2013, Bonnet et al. 2021).
In addition to mechanotransductive signaling, well geometry may affect EB development more directly. Substrate curvature has a known impact on stem cell dynamics. For instance, cells will attempt to migrate away from concave regions into convex areas (Werner et al. 2016). Round microwells are entirely concave, whereas butterfly microwells alternate between concave and convex regions. As a result, butterfly-shaped wells may prompt cells to migrate inward, while round wells incite no net movement during confinement, which could have longer-term consequences for the hCOs’ development. Curvature has also been observed to influence stem cell fates, though this effect is yet to be studied in neural stem cells (Werner et al. 2016). Although the observed phenotypic differences appeared post-confinement, it is possible that curvature-induced differences in stem cell migration and differentiation led to the disruption of rosette maintenance in butterfly hCOs. Cell migration in post-confinement organoids could be observed in further detail by using fluorescent tagging to track cell positions as these organoids develop.
Lastly, the role of naturally established morphogen gradients in the hCOs is worth considering in future investigations. The differences in microwell shapes likely result in distinct gradients of secreted morphogens, which can have a profound effect on EB development. As previously discussed, SHH exposure influences rosette maintenance. SHH gradients produced in butterfly EBs/hCOs may be uneven or restricted in magnitude compared to round EBs/hCOs. Computational modeling could be leveraged to further investigate if and how different microwell shapes alter morphogen gradients established in EBs/hCOs.
Regardless of cause, one of the most confounding aspects of our results is the fact that statistically significant differences in rosette morphology do not appear until 40 days post-confinement, at which point the hCOs have spent over a month in identical conditions. This is likely the result of less visibly apparent changes compounding over time. Future studies should attempt to pinpoint precisely where those initial post-confinement differences lie through fluorescent tracking of cell positions and/or monitoring activity of signaling pathways such as Wnt over time.
In conclusion, we found that well-geometry noticeably impacts hCO rosette maintenance. Mechanotranductive upregulation of Wnt is likely a driving factor behind the deterioration of rosettes in butterfly hCOs. However, study of other factors such as morphogen gradients and geometry-dependent cell migration are needed to fully understand the mechanisms behind these phenotypes.
Materials and Technique
Cryosectioning
We analyzed round and butterfly hCOs prepared as previously described by Sen et al. (2021), which had been fixed 20, 30 and 40 days post-confinement in a 4% agarose gel. This resulted in six sets of hCOs, each containing 3-5 hCOs. These were sectioned into 30μm thick slices using a Cryostar nx50 (Thermo Fisher Scientific) and placed onto microscope slides for storage.
Immunostaining
Samples from the different regions of the hCOs were selected and immunostained as previously described (Sen et al. 2020). Organoid tissue was stained in a solution prepared with a mixture of primary antibodies, donkey serum (Jackson Immunoresearch), Triton X-100 and phosphate-buffered saline. Secondary staining solutions were composed similarly but replacing primary antibodies with fluorescent secondary antibodies with affinities for the respective primary antibody host. DAPI nucleic acid stain (ThermoFisher) was used to non-selectively stain tissue. Phosphate-buffered saline was used to perform intermediate washing steps.
The following antibodies were used to label SOX2, TUJ1 and phospho-vimentin (pVIM):
Samples (Round and Butterfly) |
Marker |
Host |
Manufacturer |
Cat # |
Dilution |
Day 30 |
SOX2 |
Rabbit |
Millipore |
AB5603 |
1:100 |
Days 20, 40 |
SOX2 |
Goat |
R&D Systems |
AF2018 |
1:20 |
Day 20 |
TUJ1 |
Mouse |
Sigma-Aldrich |
T8578-100UL |
1:500 |
Days 20, 30, 40 |
TUJ1 |
Rabbit |
Sigma-Aldrich |
T2200-200UL |
1:100 |
Days 20, 30, 40 |
pVIM |
Mouse |
MBL International |
D076-3S |
1:200 |
Corresponding secondary antibodies were used to provide fluorescence:
Samples (Round and Butterfly) |
Marker |
Host |
Manufacturer |
Cat # |
Dilution |
Day 30 |
Anti-Rabbit Alexa Fluor 647 |
Donkey |
Abcam |
ab150075 |
1:250 |
Days 20, 40 |
Anti-Goat Alexa Fluor 647 |
Donkey |
Jackson Immunoresearch |
705605003 |
1:125 |
Day 20 |
Anti-Mouse Alexa Fluor 546 |
Donkey |
ThermoFisher |
A10036 |
1:250 |
Days 20, 30, 40 |
Anti-Rabbit Alexa Fluor 488 |
Donkey |
ThermoFisher |
A21206 |
1:250 |
Days 20, 30, 40 |
Anti-Mouse Alexa Fluor 488 |
Donkey |
Abcam |
ab150105 |
1:250 |
Microscopic imaging
Slide covers were applied to samples using ProLong Antifade Diamond mountant (Thermo Fisher Scientific). Tissue imaging was performed using a Nikon A1R microscope (Nikon Instruments) using the device’s widefield imaging mode.
Image Analysis
Captured images were manually reviewed in the image software FIJI (Schindelin et al. 2012). Individual rosettes and SOX2 aggregates were counted, and best-fitting ellipses were used to estimate rosette areas. Rosette density was measured by dividing the number of rosettes on each section by that section’s total area. This value was then averaged with those belonging to the same hCO, weighted by area. Average rosette area was determined similarly. First, areas were averaged per section, and these results were then averaged with other sections from the same hCO, weighted by area. When calculating the ratio of rosette to hCO area, empty regions within the sample’s borders (such as holes and fluid-filled sacs) were discounted. In order to exclude sections very close to the top or bottom of the hCOs, a minimum slice-area threshold of 300,000μm2 for day 20 hCOs and 700,000μm2 for days 30 and 40 was used.
Statistical Analysis
A Shapiro–Wilk test was used to determine normality. Data with a p value greater than .05 were assumed to follow a normal distribution. Where data were normally distributed, a two tailed, two-sample, unequal variance t-test was used to determine statistical significance, with a threshold of p < .05 for significance differences. Where data were not normally distributed, a Mann-Whitney U test was used with a threshold of p < .05 for significance.
Acknowledgments
We thank R. Chris Estridge for his assistance with cryosectioning and microscopy as well as for his training in FIJI. Research reported in this publication was supported by the National Institute On Drug Abuse of the National Institutes of Health under Award Number DP1DA044359, and the North Carolina State University Research Experience for Undergraduates program. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Conflict of Interest
The authors report no conflict of interest.
References
1. Zamenhof, S. and Van Marthens, E. (1974) ‘Study of factors influencing prenatal brain development’, Molecular and Cellular Biochemistry, 4(3), 157–168, available: https://doi.org/10.1007/BF01731477.
2. Briscoe, J. and Ericson, J. (1999) ‘The specification of neuronal identity by graded sonic hedgehog signalling’, Seminars in Cell & Developmental Biology, 10(3), 353– 362, available: https://doi.org/10.1006/SCDB.1999.0295.
3. Fukuchi-Shimogori, T. and Grove, E.A. (2001) ‘Neocortex Patterning by the Secreted Signaling Molecule FGF8’, Science, 294(5544), 1071–1074, available: https://doi.org/10.1126/science.1064252.
4. Georgieff, M.K. (2007) ‘Nutrition and the developing brain: nutrient priorities and measurement’, The American Journal of Clinical Nutrition, 85(2), 614S-620S, available: https://doi.org/10.1093/ajcn/85.2.614S.
5. Moore, S.W. and Sheetz, M.P. (2011) ‘Biophysics of substrate interaction: influence on neural motility, differentiation and repair’, Developmental Neurobiology, 71(11), 1090, available: https://doi.org/10.1002/DNEU.20947.
6. Sokol, S.Y. (2016) ‘Mechanotransduction During Vertebrate Neurulation’, Current Topics in Developmental Biology, 117, 359–376, available: https://doi.org/10.1016/BS.CTDB.2015.11.036.
7. Stukel, J.M. and Willits, R.K. (2016) ‘Mechanotransduction of Neural Cells Through Cell–Substrate Interactions’, Tissue Engineering. Part B, Reviews, 22(3), 173, available: https://doi.org/10.1089/TEN.TEB.2015.0380.
8. Chighizola, M., Dini, T., Lenardi, C., Milani, P., Podestà, A., and Schulte, C. (2019) ‘Mechanotransduction in neuronal cell development and functioning’, Biophysical Reviews, 11(5), available: https://doi.org/10.1007/s12551-019-00587-2. 9.
9. Saha, K., Keung, A.J., Irwin, E.F., Li, Y., Little, L., Schaffer, D. V., and Healy, K.E. (2008) ‘Substrate Modulus Directs Neural Stem Cell Behavior’, Biophysical Journal, 95(9), 4426–4438, available: https://doi.org/10.1529/BIOPHYSJ.108.132217.
10. Solanki, A., Shah, S., Memoli, K.A., Park, S.Y., Hong, S., and Lee, K.B. (2010) ‘Controlling Differentiation of Neural Stem Cells Using Extracellular Matrix Protein Patterns’, Small (Weinheim an der Bergstrasse, Germany), 6(22), 2509, available: https://doi.org/10.1002/SMLL.201001341.
11. Kijeńska, E., Prabhakaran, M.P., Swieszkowski, W., Kurzydlowski, K.J., Ramakrishna, S. (2012) ‘Electrospun bio-composite P(LLA-CL)/collagen I/collagen III scaffolds for nerve tissue engineering’, J Biomed Mater Res B Appl Biomater, 100(4):1093-102. available: 10.1002/jbm.b.32676. Epub. PMID: 22438340.
12. Chen, W., Shao, Y., Li, X., Zhao, G., and Fu, J. (2014) ‘Nanotopographical Surfaces for Stem Cell Fate Control: Engineering Mechanobiology from the Bottom’, Nano today, 9(6), 759, available: https://doi.org/10.1016/J.NANTOD.2014.12.002.
13. Sen, D., Voulgaropoulos, A. and Keung, A.J. (2021) ‘Effects of early geometric confinement on the transcriptomic profile of human cerebral organoids’, BMC Biotechnology, 21(1). available: https://doi.org/10.1186/s12896-021-00718-2.
14. Zhang, S. C., Wernig, M., Duncan, I. D., Brüstle, O., and Thomson, J. A. (2001) ‘In vitro differentiation of transplantable neural precursors from human embryonic stem cells’, Nat Biotechnol, 19(12), 1129–1133. available: https://doi.org/10.1038/nbt1201-112
15. Elkabetz, Y., Panagiotakos, G., Al Shamy, G., Socci, N. D., Tabar, V., and Studer, L. (2008) ‘Human es cell-derived neural rosettes reveal a functionally distinct early neural stem cell stage’, Genes & Development, 22(2), 152–165. available: https://doi.org/10.1101/gad.1616208.
16. Banda, E., McKinsey, A., Germain, N., Carter, J., Anderson, N. C., and Grabel, L. (2015) ‘Cell polarity and neurogenesis in embryonic stem cell derived neural rosettes’, Stem Cells and Development, 24(8), 1022–1033. available: https://doi.org/10.1089/scd.2014.0415.
17. Medelnik, J. P., Roensch, K., Okawa, S., Del Sol, A., Chara, O., Mchedlishvili, L., and Tanaka, E. M. (2018) ‘Signaling-dependent control of apical membrane size and self-renewal in rosette-stage human neuroepithelial stem cells’, Stem Cell Reports, 10(6), 1751–1765. available: https://doi.org/10.1016/j.stemcr.2018.04.018.
18. Townshend, R. F., Shao, Y., Wang, S., Cortez, C. L., Esfahani, S. N., Spence, J. R., O'Shea, K. S., Fu, J., Gumucio, D. L., and Taniguchi, K. (2020) ‘Effect of cell spreading on rosette formation by human pluripotent stem cell-derived neural progenitor cells’, Frontiers in Cell and Developmental Biology, 8. available: https://doi.org/10.3389/fcell.2020.588941.
19. Main, H., Radenkovic, J., Jin, S. B., Lendahl, U., and Andersson, E. R. (2013) ‘Notch signaling maintains neural rosette polarity’, PLoS ONE, 8(5). available: https://doi.org/10.1371/journal.pone.0062959.
20. Loerakker, S., Stassen, O. M. J. A., Ter Huurne, F. M., Boareto, M., Bouten, C. V. C., and Sahlgren, C. M. (2018) ‘Mechanosensitivity of jagged–notch signaling can induce a switch-type behavior in vascular homeostasis’, Proceedings of the National Academy of Sciences, 115(16). available: https://doi.org/10.1073/pnas.1715277115.
21. Totaro, A., Castellan, M., Battilana, G., Zanconato, F., Azzolin, L., Giulitti, S., Cordenonsi, M., and Piccolo, S.(2017) ‘Yap/taz link cell mechanics to notch signalling to control epidermal stem cell fate’, Nature Communications, 8(1). available: https://doi.org/10.1038/ncomms15206.
22. Cha, B. and Srinivasan, R.S. (2016) ‘Mechanosensitive β-catenin signaling regulates lymphatic vascular development’, BMB Reports, 49(8), 403–404. available: https://doi.org/10.5483/bmbrep.2016.49.8.112.
23. Cha, B., Geng, X., Mahamud, M. R., Fu, J., Mukherjee, A., Kim, Y., Jho, E. H., Kim, T. H., Kahn, M. L., Xia, L., Dixon, J. B., Chen, H., and Srinivasan, R. S. (2016) ‘Mechanotransduction activates canonical Wnt/β-catenin signaling to promote lymphatic vascular patterning and the development of lymphatic and lymphovenous valves’, Genes & Development, 30(12), 1454–1469. available: https://doi.org/10.1101/gad.282400.116.
24. Li, X. J., Zhang, X., Johnson, M. A., Wang, Z. B., Lavaute, T., and Zhang, S. C. (2009) ‘Coordination of sonic hedgehog and Wnt signaling determines ventral and dorsal telencephalic neuron types from human embryonic stem cells’, Development, 136(23), 4055–4063. available: https://doi.org/10.1242/dev.036624.
25. Cruciat, C. M., and Niehrs, C. (2013) ‘Secreted and transmembrane wnt inhibitors and activators’, Cold Spring Harbor perspectives in biology, 5(3), a015081. available: https://doi.org/10.1101/cshperspect.a015081
26. Bonnet, C., Brahmbhatt, A., Deng, S. X., and Zheng, J. J. (2021) ‘Wnt signaling activation: targets and therapeutic opportunities for stem cell therapy and regenerative medicine’, RSC chemical biology, 2(4), 1144–1157. available: https://doi.org/10.1039/d1cb00063b
27. Werner, M., Blanquer, S. B. G., Haimi, S. P., Korus, G., Dunlop, J. W. C., Duda, G. N., Grijpma, D. W., and Petersen, A. (2016) ‘Surface curvature differentially regulates stem cell migration and differentiation via altered attachment morphology and nuclear deformation’, Advanced Science, 4(2), 1600347. available: https://doi.org/10.1002/advs.201600347.
28. Sen, D., Voulgaropoulos, A., Drobna, Z., and Keung, A. J. (2020) ‘Human cerebral organoids reveal early spatiotemporal dynamics and pharmacological responses of ube3a’, Stem Cell Reports, 15(4), 845–854. available: https://doi.org/10.1016/j.stemcr.2020.08.006.
29. Schindelin, J., Arganda-Carreras, I., Frise, E., Kaynig, V., Longair, M., Pietzsch, T., Preibisch, S., Rueden, C., Saalfeld, S., Schmid, B., Tinevez, J. Y., White, D. J., Hartenstein, V., Eliceiri, K., Tomancak, P., and Cardona, A (2012) ‘Fiji: an open-source platform for biological-image analysis’, Nature Methods, 9(7), 676–682. available: doi:10.1038/nmeth.2019