Authors: Padhmanand Sudhakar and Prasanth Subramani
Institution: St.Peter's Engineering College, Anna University
Date: January 2007
ABSTRACT:
Malaria, unlike many diseases, has evolved as a result of millions of years of interactions at various levels with the mammalian system and hence has found ways to gather resistance to drugs and insensitivity to other treatment modalities. Co-evolution and co-adaptation of the parasite, recrudescence, recurrence, drug resistance, and the complex pathophysiology of the infection process complicate treatment regimens, most of which are ineffective. This brings us to the conclusion that vaccination is the ultimate eradicative strategy. This paper's core idea is to give an extensive overview on the latest developments of anti-malarial vaccine technology. Malaria, being a multifocal disease, is very complex on many grounds. In order to elicit proper and effective immune responses which are good enough to prevent mortality and morbidity, the parasite's multi stage pathology has been decoded, which in turn gives us a hint that many antigens are involved in establishing infection. Modern malarial vaccines, as against conventional ones, incorporate many antigens from various stages of the parasite's life cycle, so that the immune response is multi-gated. The main reason for this approach is that exposure to the whole parasite elicits a more potent, protective immune response than do the subunit vaccines tested thus far. It is more likely that immunization with only a few parasite proteins cannot duplicate the immunity elicited by exposure to a parasite that has thousands of proteins. Modern vaccines rely on the fact that if natural immunity could be mimicked, then vaccination would prevent severe malaria and malaria-related deaths. In the case of naturally acquired immunity, the complexity would be expanded by exposure to many polymorphic strains of P. falciparum. If this is the case, then the malaria genome and SNP projects may provide the essential foundation for duplicating this whole-organism immunity. Analysis of the sequence of the P. falciparum genome has dramatically expanded our knowledge regarding the paralogous expansion in the genome of parasite proteins expressed on the erythrocyte surface. In this article, we would like to address the latest research and developments on the integration of new technologies like reverse vaccinology, vaccinomics, microarrays, along with providing a better understanding of the pathogenesis of the malarial infection and the omics' revolution to herald the profiling of new candidate antigens responsible for engendering naturally acquired immunity, which will enable the formulation of protective vaccines.
INTRODUCTION:
Malaria has been one of the most prominent and ancient diseases which has been profiled and studied. It has been one of the greatest burdens to mankind, with a mortality rate that is unmatched by any other modern disease other than tuberculosis. This dreadful disease, caused by four different agents (Plasmodium falciparum, Plasmodium vivax, Plasmodium malariae and Plasmodium ovale) of the same genus, is a major health problem in most of the countries in the Tropics. Malaria affects more than 2400 million people, over 40% of the world's population, in more than 100 countries in the tropics from South America to the Indian peninsula. The tropics provide ideal breeding and living conditions for the anopheles mosquito, and hence this distribution. Over the years, as per statistical records, it has been estimated that there may be three hundred to five hundred million new infections and one to three million infection related deaths annually caused by malaria, and it has also been found that more than 90% of these deaths occur in the regions in and around Sub-Saharan Africa. Malaria kills in 1 year what AIDS killed in 15 years. In 15 years, if 5 million have died of AIDS, 50 million have died of malaria. It accounts for 2.6 percent of the total disease burden of the world. It is responsible for the loss of more than 35 million disability-adjusted life-years each year. Estimated worldwide expenditure on malaria research comes down to US$ 58 million, one thousandth of the US$ 56 billion spent globally on health research annually. Even with this overwhelming fatality and infection rates, epidemiological analyses in recent times have shown that the medical impact of Malaria may have been significantly underestimated (Breman 2001). Inspite of all this, the enormous economic impact of Malaria has never been adequately considered (Gallup and Sachs 2001). Malaria imposes a huge burden on human resources of a nation and in consequence, the Gross Domestic Product of the country gets reduced by as much as 1.3% (Gallup and Sachs 2001).
Yet, in this age of tremendous technological and medical advancements, it is a big surprise that human knowledge power has not been able to control this disease, if not eradicate it. The intolerable impact of malaria has been sometimes fully or partially attributed to the increasing resistance of the Plasmodium parasite to chemoprophylactic and chemotherapeutic agents, and the resistance of the Anopheles sp. mosquito vector to insecticides, including the pyrethroids used in insecticide-impregnated bednets. Adding to these is the inability of the health, public and civic work departments of the affected countries in mobilizing and sustaining the resources required for malarial control. The economic status of a vulnerable country plays another role in determining the equippedness and control measures in case of epidemics and endemics. For example, there has been a resurgence of malaria in areas formerly free of the disease. These complications make the disease an even more tightly knit puzzle to solve. The World Health Organization (WHO) has forecast an annual growth of 16% in malarial cases. Considering these predictions as well as the current situation at hand, it is the duty of the scientific community to focus on the prospects and possibilities of developing a vaccine for malaria. In our review, we would like to outline the recent multidimensional efforts aimed at adopting suitable methods by which natural immunity like responses can be generated by a malarial vaccine. The pith of this work is to give a precise description on the various parasitic and host factors which are taken into consideration in modern malarial vaccinology.
PATHOPHYSIOLOGY AND LIFECYCLE OF THE MALARIAL PARASITE:
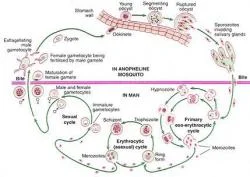
Figure 1. Schematic Representation of the Malarial Parasite's Life Cycle.
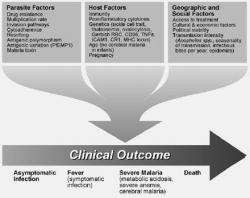
The life cycle of the malarial parasite is very complex (Figure 1). P. falciparum and, to a much lesser extent, P. vivax (Mendis et al 2001) are the main causes of disease and death due to malaria. Mosquitoes inject parasites in the form of sporozoites into the subcutaneous tissue and less frequently, directly into the bloodstream and from there, they travel to the liver. Recent evidence indicates that sporozoites pass through several hepatocytes before invasion is followed by parasite development (Mota MM et al 2001). The co-receptor on sporozoites for invasion involves, in part, the thrombospondin domains on the circumsporozoite protein and on thrombospondin-related adhesive protein (TRAP). These domains bind specifically to heparin sulfate proteoglycans on hepatocytes in the region in apposition to sinusoidal endothelium and Kuppfer cells (Frevert et al 1993). Within the hepatocyte, each sporozoite develops into tens of thousands of merozoites, each able to invade a Red Blood Cell (RBC) on release from the liver. Disease begins only once the asexual parasite multiplies within the RBC. This is the only gateway to disease. P. falciparum and P. vivax within the RBC develop over 48 hours, producing around 20 merozoites from a mature parasite, each able to invade other RBCs. A small proportion of asexual parasites converts to gametocytes that are critical for the transmission of the infection to others through female anopheline mosquitoes but cause no disease. Here the strategy of P. vivax differs from that of P. falciparum. P. vivax develops into gametocytes soon after release of merozoites from the liver, whereas P. falciparum gametocytes develop much later. Early treatment of clinical malaria attacks by anti-bloodstage chemotherapy for P. falciparum also kills the developing gametocytes and in the case of P. vivax, it transmits before the symptomatic stage of the disease. The outcome of the infections differs from person to person and this is another manifestation of the highly variant factors (Figure 2) involved in malarial infection and disease.
Figure 2. Factors affecting the ultimate disease outcome in Malarial infection.
HOW ARE RED BLOOD CELLS PLAGUED BY THE MALARIAL PARASITE?
The sequence of invasion is probably similar for all Plasmodium spp. The parasite must engage receptors (Chitnis 2001) on RBCs for binding and undergo apical reorientation (Dvorak et al 1975) junction formation (Aikawa et al 1978) and signaling. The parasite then induces a vacuole derived from the RBC plasma membrane and enters the vacuole by a moving junction. Three organelles on the invasive or apical end of parasites (rhoptries, micronemes, and dense granules) define the phylum Apicomplexa. Receptors for invasion of RBC by merozoites and for invasion of liver by sporozoites are found in micronemes (Adams et al 1990), on the cell surface, and in rhoptries. The distribution of these receptors within an organelle may protect the parasite from antibody-mediated neutralization, as the release after contact with the RBC may limit their exposure to antibody. A critical question in parasite biology remains the identification of the signaling pathways to release organellar contents in contact with a host RBC. Malarial parasites have intracellular signaling pathways through phosphoinositide, cyclic AMP (cAMP), and calcium-dependent pathways. What remains completely unknown is which merozoite surface molecules recognize the RBC surface and signal for the invasion process. These events include release of critical molecules from apical organelles and the initiation of the actin-myosin moving junction that brings the parasite within the vacuole that forms in the RBC. The TRAP protein interacts with skeletal proteins in malarial sporozoites and in Toxoplasma gondii (Kappe et al 1999), but the equivalent molecule for merozoites has yet to be identified.
Both P. falciparum and P. vivax can cause severe anemia, but only P. falciparum causes the multiple complications of cerebral malaria, hypoglycemia, metabolic acidosis, and respiratory distress. Certain differences in the biology of the two parasites partially explain the differences in patterns of disease. First, P. falciparum can invade a large percentage of the RBCs, whereas P. vivax is limited to reticulocytes. Similar differences are found between virulent and avirulent P. yoelii. Both invade reticulocytes preferentially, but once the reticulocytes are consumed, virulent P. yoelii can invade all RBCs, leading to higher parasitemia and death. A recent study by Chotivanich of severe malaria compared to uncomplicated falciparum malaria suggested a similar pattern, with the virulent P. falciparum invading all RBCs and the avirulent parasites invading only a subpopulation (Chotivanich et al 2000). A second difference is the surprising redundancy of invasion pathways in P. falciparum, lacking in P. vivax. P. vivax invades only Duffy blood group-positive RBCs (Miller et al 1976) and is largely limited to reticulocytes. In West Africa, where the Duffy blood group is missing on the RBCs, P. vivax essentially disappeared. The Duffy negative blood group has also occurred independently in Papua New Guinea (Zimmerman et al 1999), an area of high endemicity of P. vivax. The limitations in invasion of P. vivax have led to the discovery of two families of parasite receptors: i) the parasite molecule that binds to the Duffy blood group system and Duffy binding-like (DBL) homologous proteins of P. falciparum and P. knowlesi (Adams et al 1992), and ii) the parasite reticulocyte-binding proteins of P. vivax (Galinski et al 1992) and reticulocyte binding-like (RBL) homologous proteins of P. falciparum (Rayner et al 2001) and P. yoelii (Keen et al 1994). The various members of the DBL and RBL families may recognize different RBC receptors than the Duffy blood group or the receptor on reticulocytes. The receptor grouping into DBL and RBL refers to the family of homologous parasite proteins, not the specificity on the RBCs for binding.
There is a large family of RBL genes in P. yoelii. Each of the merozoites within a single infected RBC can express a different member of the RBL family (Preiser et al 1999). If each has a different RBC-binding specificity, then the parasite has a greater chance for survival. Thus, although the full details for the DBL and RBL families are unknown, they clearly determine much of the flexibility for invasion by the various Plasmodium spp. This flexibility, in turn, determines the maximum parasitemia and disease caused by the various parasites.
P. falciparum can use its multiple redundant alternative pathways to invade RBCs lacking a particular receptor such as sialic acid (Okoyeh et al 1999) at equal or reduced efficiency. Three sialoglycoprotein-dependent pathways involving RBC and parasite co-receptors have been identified: i) glycophorin A and the parasite DBL protein, EBA-175 (Sim et al 1994) ,ii) glycophorin C/D and the DBL parasite protein, BAEBL (Mayer et al 2001), and iii) a trypsin-resistant pathway involving a P. falciparum RBL protein (Rayner et al 2001). A fourth may involve sialic acid on glycophorin B (Rayner et al 2001). Despite markedly reduced invasion of glycophorin A-negative RBC, only glycophorin B mutations occur in Africa. Gerbich RBCs fail to express glycophorin D and express an altered glycophorin C and have reduced binding to the parasite molecule, BAEBL. Gerbich RBCs are rare in most parts of the world except in the falciparum-endemic regions of Papua New Guinea, where the allele frequency approaches 50% (Patel et al 2001). Such redundancy and alternative pathways are a major advantage to the survival of P. falciparum in response to changes in host genetics. The parasite, however, may become less virulent as it adapts to survival in these deficient RBCs.
Studying the DBL and RBL families has begun to yield a molecular understanding of the diverse invasion pathways for P. falciparum and other Plasmodium spp. Although other parasite proteins on the merozoite surface and in apical organelles have been proposed as receptors (Patino et al 1997, Hodder et al 1996 and Triglia et al 2000), there is no direct evidence so far. Because invasion is such a complex series of events from RBC binding, to apical reorientation, to entry, that it seems likely that multiple proteins are involved for efficient invasion. For example, evidence has been presented that invasion requires cleavage of a RBC surface protein by a parasite serine protease (Braun-Breton et al 1992). This parasite enzyme has yet to be identified. Thus, the molecular and cellular events surrounding each step in invasion still remain to be elucidated. Understanding these pathways will give insight into parasite virulence and will facilitate rational vaccine design against merozoite invasion.
ANCHORING OF INFECTED RBC'S TO THE VASCULAR ENDOTHELIAL CELLS:
An important difference between P. falciparum and other human malarias is the way that P. falciparum modifies the surface of the RBC for adherence of both asexual parasites and gametocytes to the endothelium and asexual parasites within placenta. As a result, only ring forms of P. falciparum are found within the circulating blood (Baruch et al 1999). The surface of P. falciparum trophozoite- and schizont-infected RBC is covered with knob-like excrescences that are the contact points with host cells. The adherence protects the parasite from destruction, as non-adherent mature parasitized RBCs are rapidly cleared within the spleen. Trying to decipher the highly complex and pathogenic adhesion process emphasizes how much we have learned and how little we understand. To answer whether and how sequestration can lead to pathogenesis, we should first look at how the parasite sequesters. The P. falciparum adhesion process is comparable to adhesion of leukocytes, where most parasites first tether, then roll, before becoming firmly adherent (Cooke et al 1994 and Ho et al 1999). Most host receptors are involved with tethering and rolling but are unable to support firm adhesion under flow on their own (Cooke et al 1994). Binding to these host receptors is important, as it significantly increases adhesion that may allow the parasite to efficiently bind to endothelium of various organs (Yipp et al 2000). Only two receptors, CD36 and chondroitin sulfate A (CSA), provide stable stationary adherence (Cooke et al 1994).
Parasites sequester in various organs including heart, lung, brain, liver, kidney, subcutaneous tissues, and placenta. The various endothelial cells in these organs and syncytiotrophoblasts in placenta express different and variable amounts of host receptors. To successfully adhere to these cells, the parasite binds to a large number of receptors (Baruch et al 1999). The adhesion phenotype is not homogenous, and different parasites can bind to variable numbers and combinations of host receptors (Beeson et al 1999). This variability is believed to affect the tissue distribution and pathogenesis of parasites. Amazingly, a single parasite protein, the P. falciparum erythrocyte membrane protein 1 (PfEMP1) expressed at the infected erythrocyte surface, mediates its binding to the various receptors. PfEMP1 is encoded by the large and diverse var gene family that is involved in clonal antigenic variation and plays a central role in P. falciparum pathogenesis (Baruch et al 1995). The multiple adhesion domains located at the extracellular region of PfEMP1 can simultaneously recognize several host receptors. These domains contain five different DBL domains, so named for the homology to the DBL domains involved in RBC invasion and 1-2 cysteine-rich interdomain regions (CIDR) (Su et al 1995). The binding domains for several host receptors were recently mapped to various DBL and CIDR domains. The diversity within this gene family is extensive, and numerous var genes appear in the parasite population. Although each parasite expresses a single var gene (Chen et al 1998), this can change at a rate of up to 2% per generation (Roberts et al 1992).
In most cases, the binding to host endothelium does not lead to pathogenesis, as most infections result in malaria that is devoid of complications. What leads to the transition from uncomplicated to a serious infection such as cerebral malaria is unclear at present. An intriguing possibility is that expression of particular binding properties will lead to distinct patterns of sequestration and to pathogenic consequences. One example is the sequestration of infected RBC within the placenta that causes premature delivery, low birth weight, and increased mortality in the newborn and anemia in the mother. Parasitized RBCs isolated from placentas have a unique adhesion property different from parasites collected from non-pregnant individuals. These parasites bound to CSA and failed to adhere to CD36, the critical host receptor for sequestration in microvasculature. The apparent dichotomy in adhesion to these receptors was selected to allow the parasite to sequester not to endothelium but in placenta, perhaps a site of reduced immunity. Indeed, CSA-binding parasites express PfEMP1 with a DBL domain that binds CSA and a non-CD36-binding CIDR1 (Gamain et al 2001). In contrast, CD36-adherent parasites express a PfEMP1 with a CD36-binding CIDR1 (Gamain et al 2001 and Chen et al 2000).
How adhesion progresses to pathology is a major question that remains unresolved. Several mechanisms that might cause damage to host endothelium and organs have been proposed, including obstruction of blood flow and systemic or local production and deposition of proinflammatory cytokines. Parasite adhesion can also affect the endothelium by inducing or blocking signal transduction mediated by host receptors such as CD36. The recent advances in adhesion research will hopefully provide leads for the mechanism of adhesion related pathogenesis.
IMMUNE RESPONSES AGAINST THE MALARIAL PARASITE:
Antibodies and the proinflammatory response (Figure 3) protect against the asexual blood stages of some rodent malarias and probably also human malaria. The protection mediated by the proinflammatory response may relate to the cytokines TNF- and IFN- and the release of mediators such as nitric oxide (NO). Clark proposed that mediators, especially NO, are also central to disease (Clark et al 1999). It is perfectly logical that these are involved in bone marrow suppression and cerebral malaria, but the data are lacking to prove this role. Furthermore, no model exists for the study of cerebral malaria. One hypothesis suggests that TNF- induces endothelial cells from brain to express ICAM-1 (Wong et al 1992) as vessels in the brain have increased expression of ICAM-1 in cerebral malaria. Although NO has been proposed as the cause of cerebral malaria, NO is at higher levels systemically in uncomplicated malaria than in cerebral malaria (Levesque et al 1999). Coma could be caused by local increases in NO in the brain and not increased levels in blood, but this has not been measured. Indeed, total nitrate plus nitrite levels in the CSF of children with cerebral malaria are low, and it has been suggested that this may exacerbate n-methyl-D-aspartate-mediated neurotoxicity due to excitotoxins such as quinolinic acid.
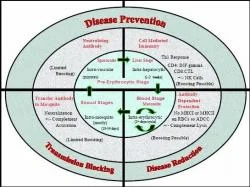
Figure 3. Immune Responses elicited during different stages of the Malarial parasite's life cycle.
Data suggesting that a toxin of malarial origin drives the proinflammatory response are interesting, but the physiologic significance is unproven at present. The evidence that a particular molecule is involved in induction of the proinflammatory response was developed by an assay for the release of TNF- by macrophages in vitro. Isolation of subcellular components from the parasite and the in vitro assay led to the identification of the GPI anchor from parasite proteins MSP1 and MSP2 as an inducer of proinflammatory cytokines. Antibodies to the GPI anchor were associated with lack of disease in adults, but proof that this is causally related is lacking. Modifications in the immune response to malaria that may not be malaria specific have been identified. Infection with P. falciparum causes apoptosis of mononuclear cells in infected humans (Naik et al 2000). In an animal model, infection with a rodent malaria to which they had previously been exposed led to apoptosis of T cells immune to malaria and not those immune to ova, a malaria-unrelated antigen. The cells that were eliminated were proinflammatory T cells, producing IFN- and IL-2, but not IL-4.
COMPLICATIONS IN MALARIAL INFECTIONS:
Malaria is one disease which complicates itself so much that common modes of treatment and prophylaxis (Desjardins et al 1988) cannot be drawn on a single level basis. Instead, a case-by-case approach must be undertaken. This might not be practically feasible as of now, though some progress has been made in deciphering the common mechanisms leading to these intricate pathological manifestations. Two such factors which significantly affect the occurrence and severity of the disease are Recurrence and Resistance.
Recurrence:
Recurrence is a problem which keeps the disease alive in a community and makes its control quite difficult (Omonuwa et al 2000). Absence of effective immune response is one of the many reasons for the recurrence phenomenon. Acquired immunity develops after several attacks of malaria over several years. This immunity is stage specific, species specific and 'strain' specific. It does not have any sterilizing effect on the parasitemia and immune adults are still re-infected. However, the clinical signs and symptoms are reduced with development of clinical immunity. various explanations have been offered for these observations. Generalized immunosuppression by the parasite, presence of T-cell independent antigens which fail to induce immune response, inhibition of B-cell proliferation, and extreme polymorphism or clonal variation of immunologically relevant antigens could be the causes for the poor immune response in malaria (Sokhna et al 2000 and Neva et al 1970).
Secondly, exposure to repeated mosquito bites and re-infection is the most profiled cause for recurrence of malaria. The patient is exposed to fresh mosquito bites soon after completing the anti malarial treatment. There is no immunity from the past infection and to worsen the situation, the antimalarials do not prevent establishment of the fresh infection. Therefore, unless the patient observes stringent anti mosquito measures, re-infection is inevitable in an endemic area (Owusu-Agyei et al 2002).
Non compliance to the treatment regimen combined with incomplete and improper treatment methods will make the patient pay heavily. All blood schizonticidal drugs suppress the erythrocytic schizogony and give symptomatic relief. The patient may stop the drugs due to various reasons like vomiting, ignorance and financial constraints. This results in recrudescence within a short period (Fahmy et al 1992). Also, if the patient does not take the complete course of primaquine, relapses can occur from re-activation of hypnozoites in the liver. Proper patient education regarding the complete course of anti malarials is very important. In cases of P. vivax and P. ovale infections, recurrent attacks could be due to re-activation of hypnozoites in the liver. This can occur any time after 30-180 days from the primary attack (Kondrachaine et al 1999). The relapses have the characteristic symptoms of malaria. Splenomegaly may be a prominent feature in these patients. Such long-term relapses commonly occur in patients who have either not taken primaquine or taken incomplete treatment.
Finally, in P. falciparum and P. malariae infections, the parasites can remain in the blood for months or even years and cause recurrent symptoms from time to time. In falciparum malaria, such recrudescence can occur within 28 days of the primary attack and may indicate partial resistance to chloroquine. However, treating every case of recurrent P. falciparum as resistant malaria is unjustified. One should consider the possibility of re-infection in most of these cases.
Resistance:
Resistance to antimalarial drugs is proving to be a challenging problem in malaria control in most parts of the world. Since early 1960s the sensitivity of the parasites to chloroquine, the best and most widely used drug for treating malaria, has been on the decline. Newer antimalarials were discovered in an effort to tackle this problem, but all these drugs are either expensive or have undesirable side effects. Moreover after a variable length of time, the parasites, especially the falciparum species, have started showing resistance to these drugs also (Lqbal et al 2002). The important factors that are associated with resistance are longer half-life, single mutation for resistance, poor compliance, host immunity, and number of people using these drugs. The characteristics of a drug that make it vulnerable to the development of resistance are a long terminal elimination half-life, a shallow concentration-effect relationship, and mutations that confer marked reduction in susceptibility. Drug resistance is most commonly seen in P. falciparum. Resistance to chloroquine is most prevalent, while resistances to most other antimalarials like pyrimethamine, quinine, mefloquine, artemesin and quinoline (Ward et al 1997) compounds have also been reported. These developments further justify the cause and urgency for formulating an effective vaccine against malaria.
PRIMARY REQUIREMENTS OF MALARIAL VACCINES:
From experience, it is learnt that different populations demand different types of malarial vaccines making use of the different arms of the natural immune response system (Figure 3). The basic purpose of a vaccine is to reduce the incidence of severe malaria, and malaria-associated mortality in infants and children with heavy exposure to Plasmodium falciparum, such as those living in sub-Saharan Africa. This constitutes a type 1 vaccine.
At the other extreme is the requirement to prevent all clinical manifestations of malaria in individuals from areas with no exposure who travel to regions where malaria is endemic, primarily malaria caused by P.falciparum and P.vivax. The vaccine fulfilling this extreme requirement is the type 2 vaccine. This extreme approach to Malaria Vaccine Development does not take into account specifically populations affected by malaria that fall between these extremes, such as individuals in endemic regions at high risk of P.vivax infections (Mendis et al 2001). In fact, as type 1 and type 2 vaccines are developed, they will need to be assessed in many different populations.
The problem is complicated by the varied epidemiology of the disease for the type 1 vaccine which is designed to reduce mortality among children in sub-Saharan Africa. In areas with extremely intense transmission, it is primarily infants who are dying of malaria, with severe anemia as the major cause of death. In areas with less intense transmission, it appears to be 2 to 5 year olds who are at risk of dying with cerebral malaria (Mendis et al 2001). It is a definite possibility that different vaccines, tailored to the predominant pathophysiology of the parasite in a particular region, or different vaccination strategies, targeting the age groups at greatest risk may be the most effective response to this heterogenous epidemiology.
Similarly, the target populations are also varied for type 2 vaccine designed to prevent all clinical manifestations of malaria in non-immune travelers to malaria-endemic areas. It is a general conception that the major recipients for this type of vaccine would be travelers from highly developed malaria-free-countries, but there are hundreds of millions of people living in non-malarious areas of malaria endemic countries, who travel to malarious areas of their own country. Surprisingly, there is little mention in the malaria literature of the increasing number of non-immune individuals living in countries with endemic malaria, who must receive short-term protection against malaria by a vaccine. Because of their susceptibility to the rapidly developing severe disease, and because of their brief exposure to transmission, it is generally thought that these in-country travelers, and their children, would require a vaccine with the same protective profile as a vaccine for travelers from highly developed malaria-free countries.
Recently, a type 3 vaccine has also been developed for the sole purpose of blocking transmission. This type of a vaccine is not designed to protect the immunized individual but the entire community by reducing transmission intensity. Such a vaccine or its components can be combined with a type 1 or type 2 vaccine to lead to what will be called a multipurpose' vaccine. Such a vaccine unquestionably would be of great value on islands with malaria, and in areas with only modest transmission. It might also be useful during prolonged epidemics. This cocktail vaccine could be quite useful, particularly when combined with vaccines targeting other parasite stages, for preventing a selective process favoring the emergence of rapidly growing and therefore virulent parasites (Gandon et al 2001). Even if not able to impact overall transmission levels, a transmission blocking component included in a type 1 or 2 vaccine could block the escape resistant mutants, thus preserving vaccine efficacy in the community and also preventing the emergence of virulent parasite strains. Thus the general idea is to block transmission along with eliciting protection against the disease.
An ideal malaria vaccine would prevent all infection by priming the immune system to destroy all parasites, whether free swimming in the blood, while in the liver, or even, theoretically, while in red blood cells. This degree of protection would be extremely difficult to achieve and might not be technically feasible with current vaccinology art and science. Many vaccine developers have therefore focused their efforts on creating a vaccine that limits the ability of the parasite to successfully infect large numbers of red blood cells. This would not prevent infection but would limit the severity of the disease and help prevent malaria deaths.
An Ideal Malarial Vaccine:
An ideal malarial vaccine will have a range of properties satisfying many conditions towards which future research efforts will be directed at. So it is necessary to define the characteristics of an ideal, virtual malarial vaccine. The ideal malarial vaccine will be highly effective in all age ranges, that is, it must elicit protective immunity in infants, children and adults. It has to be immunogenic and at the same time should confer safety and not have any undesirable side effects. In addition to being immunogenic, it should have the property of engendering long-term immunity, i.e; it should not require boosting to supplement itself. The ideal vaccine should go hand in hand with existing immunization programs and schedules and ought to be easy to administer. It should be devoid of other complications such as interference with traditional childhood vaccines. Last, but not the least, the vaccine should be easy and inexpensive to manufacture and be affordable in low-resource settings.
STUDIES RELATING TO WHOLE PARASITE ELICITED IMMUNITY:
Children who survive to the age of 7-10 years rarely develop life-threatening P.falciparum infections and rarely die of malaria in areas of sub-Saharan Africa, which has the most intense malaria transmission rates as well as the highest rates of malaria associated mortality and morbidity. This acquired immunity occurs more rapidly, the more intense the transmission of P.falciparum. Older children and adults become infected frequently, but it is thought that their immune systems limit the infections, thereby preventing severe disease. A vaccine that duplicates naturally acquired immunity and turned infants and children into 15-year olds with respect to protective immunity would essentially eliminate severe disease and death without eliminating infection or mild to moderate disease. It was found that mice immunized with radiation-attenuated rodent malaria sporozoites were protected against challenge (Nussenzweig et al 1967). Based on these rodent studies, human trials were conducted subsequently and it was reported that exposure of humans to the bite of irradiated mosquitoes carrying P.falciparum sporozoites in their salivary glands protected these volunteers (Clyde et al 1973, Rieckmann et al 1979 and Romero et al 1989). These studies have since been repeated more systematically (Edelman et al 1993, Egan et al 1993, Herrington et al 1991 and Hoffman et al 2002).
Among 14 volunteers exposed to greater than 1000 infected, irradiated mosquitoes and first challenged 2-10 weeks after the last exposure, 13 of the 14 were completely protected against infection. It was also proved that there is apparently little or no genetic restriction of the protective immunity elicited by this immunization approach. Six volunteers were challenged 23-42 weeks after the last boost and 5 of the 6 were protected. A total of seven challenges have been done with strains of P.falciparum different from the one used of for immunization, and there was protection in all the seven individuals. To combine all studies, among volunteers exposed to at least 1000 infective bites and challenged within one year after the last exposure, 33 of the 35 challenges were associated with complete protection. Thus, the irradiated sporozoites vaccine protects greater than 90% of recipients against experimental challenge for at least 10 months, and the protection is not strain-specific. A vaccine encompassing such properties will prove to be as good as both type 1 and type 2 vaccines.
CHALLENGES IN FORMULATING AN EFFECTIVE MALARIAL VACCINE:
The development of a malarial vaccine requires more than understanding the molecular, pathogenic, parasitic and epidemiologic aspects of the plasmodium species. A successful vaccine strategy requires an appreciation of how the human host immune response interacts with the parasite. Such an understanding provides vaccine developers with clues on how best to design products that stimulate the proper effector arm of the immune system to achieve the desired goal. Despite the knowledge that our immune system can control malaria, there is currently no vaccine in the market against this dreaded disease. It is not possible to consider immunizing people by exposure to live parasites, which is what occurs in naturally acquired immunity, as the individuals would become ill. It has not been practical to consider immunization by the bite of irradiated, infected mosquitoes or by intravenous injection of live-radiation attenuated sporozoites to duplicate the immunity elicited by the irradiated sporozoites vaccine. The approach has been to try to determine the immune responses responsible for the protective immunity seen in these two models, and the parasite life cycle stages and proteins, against which this protective immunity is directed, and then to develop vaccine delivery systems that would induce the required responses against the identified targets.
Current and future advances in understanding human immunology and the biology of the malarial parasites, many of which will be dependent on the data from the human genome sequencing projects (Lander et al 2001 and Venter et al 2001) and the malaria genome projects (Bowman et al 1999, Gardner et al 1998 and Gardner et al 1999), should allow the identification of key antigens associated with the protection and the formulation of vaccines effective in all recipients, regardless of their genetic background. The availability of the entire P.falciparum genome has provided a major boost in increasing the number of identified candidate antigens and has significantly brightened the prospects of developing an effective malarial vaccine.
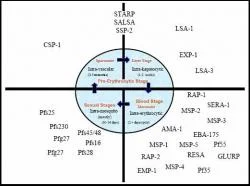
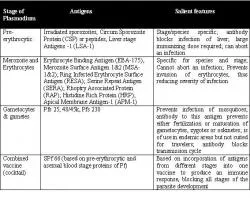
Figure 4. Stage-wise distribution of the antigenic profile of the Malarial parasite.
Parasitic Complexity:
Malarial parasites are much more complex than other microbes such as viruses or bacteria which have been controlled extensively by vaccination. The complexity of the malarial parasites is reflected by their life cycle which has many stages, each of which is characterized by the expression of different and unique proteins (Figure 4). When sporozoites are inoculated into humans by Anopheles mosquitoes, they circulate extracellularly in the bloodstream for less than 30 minutes before entering the liver. Within the hepatocyte, a uninucleate sporozoite develops into a schizont with estimated 10000 - 40000 uninucleate merozoites.
These merozoites rupture from the hepatocyte and each can invade an erythrocyte, initiating the cycle of intraerythrocytic stage development, rupture and reinvasion that leads to a 10-20 fold increase in the number of parasites in the bloodstream every 48 hours. These asexual erythrocytic stage parasites are responsible for the clinical manifestations and pathology of malaria. Erythrocytic stage sexual parasites, called gametocytes are ingested by mosquitoes, in which they develop into sporozoites over a period of 14-21 days. The proteins expressed by each of these various stages are antigenically distinct. Thus, if a vaccine elicits high levels of anti-sporozoite antibodies, those antibodies will generally not recognize the asexual erythrocytic stages that follow. Furthermore, for many of these genes and proteins, allelic and antigenic variation has been demonstrated. A single individual can be infected simultaneously with at least eight different strains (Felger et al 1999), which may vary at critical B and T cell epitopes. This allelic heterogeneity is expanded by extensive antigenic variation present within a single strain.
For example, PfEMP1, a protein expressed on the surface of erythrocytes, is encoded by 50-100 different genes, each with some variation in its sequence (Patrick Duffy et al 2001). The parasite probably expresses only one at a time, with each new variant escaping from antibodies induced by the previous variant and therefore creating a new wave of parasitemia. PfEMP1 is thought to mediate cytoadherence of infected erythrocytes to endothelial cells in the microcirculation during maturation of the parasite, thereby preventing removal of the infected erythrocyte in the spleen during this vulnerable phase of the cycle. Cytoadherence is thought to be responsible for the microcirculatory obstruction important to the pathogenesis of severe disease. In summary, stage-specific expression of proteins, the presence of multiple antigenically distinct strains in nature, and within-strain antigenic variation are critical to the parasite's survival, unfavorable to the host, and greatly complicate the challenge for vaccine developers.
New strides in research have been undertaken to develop subunit vaccines that provide protective immunity comparable to that of the human models of naturally acquired immunity and radiation attenuated sporozoite immunization. However, no such vaccine has provided comparable protection.
There are many reasons for the lack of success in developing a subunit vaccine which duplicates naturally acquired immunity. One of the reasons is that exposure to the whole parasite elicits a more potent, protective immune response than the subunit vaccines tested so far. However, in the case of people protected by the irradiated sporozoite vaccine or by naturally acquired immunity, T-cell and antibody responses against our current candidate antigens (Berzins et al 1996, Brown et al 1996, Hoffman et al 2002, Holder et al 1996, Mbogo CN et al 1995 and Sinnis P et al 1996) or, in the case of antibody responses, against whole parasites are generally modest at best and in many instances lower than those achieved by subunit vaccination. It is more likely that immunization with only a few parasite proteins cannot duplicate the immunity elicited by the exposure to a parasite that has thousand of proteins: modest immune responses against tens, hundreds or thousands of parasite proteins may be additive or synergistic and may underlie the protection observed in these human models. In case of naturally acquired immunity, this "breadth" would be expanded by exposure to so many polymorphic strains of P.falciparum. In summary, whole-parasite induced immunity could be directed at many of the 5000-6000 malaria parasite proteins. Knowledge of all these potential targets and their variability at the epitope level has been documented a great deal after the publishing of the malarial parasite's genome and also the SNP (Single Nucleotide Polymorphisms) profiles of the genes coding for many of the potential targets.
Tasking the field trials:
Clinical trials are primary manifestations of the scientific methodologies employed in medical pharmacology. Subjectal and anecdotal experiences have been replaced by scientific methods providing greater and greater objectivity, rigor and efficiency in assessing the impact of a vaccine. Currently, the task is to get enough potential malaria vaccines to and through the series of clinical trials needed to bring to the world a licensed product. Vaccine field trials are long-term studies aimed at assessing the safety, efficacy and immunogenicity of a new vaccine product. Sometimes trials also assess how well the product in question meshes with existing healthcare delivery systems, such as national immunization programs. Field trials and their organization continue to be very critical factors involved in confirming vaccine efficacy and acceptability. Clinical trials are carried out in phases and each phase informs the developers about the next steps of testing and development. The full set of clinical trials for a successful candidate can take more than 10 to 12 years and may involve as many as 100,000 volunteers and may cost upto $500 million. In this regard, the onus will be to decide which outcome variables to measure in field trials and which populations to study. Primarily, trials and testing should not result in mortality and disease of any degree. There is a potential problem that the vaccine may be discarded as a result of initial studies because the proper outcome variables were not measure appropriately. It will be difficult to use severe disease and death as the primary outcome variables in initial studies because of the very large sample sizes required.
It is very important to identify the groups at the highest risk so that the sample sizes can be reduced. Current investigations seek to identify surrogates of severe disease and death. These surrogates can be of different forms like parasitological, hematological, biochemical and clinical manifestations that are predictive about the disease outcome. Several other areas of field research could provide data to aid vaccine development. The most important of these is the identification of target groups for vaccines in different areas and the exclusion of groups, like those with sickle-cell trait, who are at decreased risk, and do not need to be immunized. It is important to determine whether there are measurable outcome variables that have a high predictive value for severe disease and malaria-associated mortality.
As discussed before, ideal vaccines are just virtual benchmarks which will be used as references for optimization. While a less-than-ideal vaccine is expected to have some significant public health impacts, lack of some desired characters could affect how and where the vaccine is implemented. For example, if annual or biannual booster doses are necessary, there will be an increased burden on the healthcare system. Or, if the best product limits the severity of the infection and reduces morbidity and mortality but does not prevent all disease, governments and individuals will have more to consider in deciding whether, when, and how to use the vaccine. This is why carefully designed and executed vaccine trials are so important. They actually help us in understanding the strengths and weaknesses of each vaccine formulation, and guide the way to the best, if not the ideal vaccine.
The Overwhelming need to redefine immunological indicators/variables of protection:
Most existing vaccines are thought to provide protection primarily by the induction of protective, neutralizing antibodies. The humoral response can easily be assessed. Malarial vaccines are thought to have multiple and complex mechanisms of protection, including antibodies and cell-mediated immunity. Malarial infection comes with biggest problem that it has a strong possibility of reactivation following a temporal period of relapse. So malarial vaccines must confer long-lasting immunity which must prevent infection and disease. At the same time, we must recognize that it may be unreasonable to expect the degree of protection with new vaccines that we have taken for granted in the current childhood vaccines.
All these arguments bring us to the point of realization of absolute truth, which is that we need to understand the mechanisms of immunity and the responses that are necessary to engender protection. This thrust for knowledge begins with identifying correlates of protection and the immunological measures and tests that appear to parallel protection. Ultimately, such correlates or variables that are firmly established will be assessed in clinical trials as surrogates for immunity which translate into laboratory tests that predict protection with high accuracy. Different immune responses may be required for different forms of the parasite. For example, there are antibodies which have been shown to prevent mosquito-derived sporozoites from infecting liver cells, those that kill the blood forms, and those that kill the gametocytes. There has been a lot evidence to show that there are specific cytotoxic T-cell Lymphocytes that kill infected liver cells. These could be assayed in vitro, but the most challenging part in vaccine efficacy evaluation is to decide and finalize a set of the above mentioned correlates which will indicate protective immunity elicited by the vaccine. So, one of the major visions of malarial vaccine research is to employ the best methodology and technology available in clinical trials and in animal models to analyze those immune responses that correlate best with protection. These correlates and surrogate end points will accelerate the period of clinical testing, reduce costs, and allow more vaccine candidates to be studied and analyzed.
IMMUNOPATHOLOGICAL BASIS OF VACCINE DESIGN:
Design and formulation of vaccines are always preceded by a thorough investigation of the immunopathological states and infection modes of the parasite. Parasites have to survive in their vertebrate host during a sufficiently prolonged time in order to achieve their life cycle through successful transmission via insect vectors. In their vertebrate hosts, parasites are often confronted by vigorous effector immune responses that they must subvert somehow in order to survive and be transmitted successfully. The immune response comprises several components in terms of effector cells, antibodies, and signalling molecules such as cytokines. It is now well-recognized that not all components of the immune response triggered during the process of parasitism have anti-parasitic activities or properties. In this context, one of the strategies devised by many parasites has been to utilize some components of the host immune response to its own benefit. Therefore, an effective vaccine against intracellular pathogens should only induce effector mechanisms ultimately leading to the destruction of the parasites. The vaccine should abstain from triggering immune components of the immune response favoring the survival of the parasites.
Parasites have also devised a variety of ways by which to escape the effector components of the immune response that they have elicited in their mammalian hosts. Therefore, the balance between the components of the immune response with effector functions and those irrelevant for protection on the one hand, and the effector immune response and the escape processes induced by parasites on the other largely determine the outcome of the infection. Thus, elucidation of the mechanisms involved in the progression of parasitic diseases and definition of the relevant immune effector mechanisms are equally important for the identification of potential targets for beneficial intervention and subsequent vaccinisation. Indeed, the specific triggering of potent effector mechanisms and responses without activating the immune responses' components leading to pathology, together with the prevention of the escape mechanisms evolved by parasites, are the aims of vaccinisation.
The importance of immunoregulatory molecules like cytokines in the immunopathology of diseases is worthy enough to be mentioned. Their central role as antiparasite factors and as mediators of pathology has been reinforced by the description of two functionally different CD4+ T cell populations that can be distinguished on the basis of the patterns of cytokines which they produce. In both mice and humans, Th1 cells produce Interleukin-2 (IL-2) and Interferon gamma (IFN gamma), whereas Th2 cells produce IL-3, IL-4, IL-5, IL-10, and IL-13. Th1 cells are responsible for cell-mediated immune reactions, whereas Th2 cells are involved in humoral immunity. Resistance and susceptibility to several infections by many pathogens have been analyzed with respect to the Th1-Th2 paradigm. Resistance to intracellular pathogens is often associated with the development of a polarized Th1 response. Although resistance to extracellular parasites is now known to correlate with the generation of Th2 cells, the respective roles of Th1 or Th2 effector responses in protective immunity against many diseases like Schistosoma, an extracellular pathogen, are not yet fully understood. Furthermore, expression of immunity in some parasitic diseases such as malaria cannot simply be analyzed in view of the development of polarized Th1 or Th2 responses because infection with this parasite is a multifocal process.
There is very little doubt that immune responses against Plasmodium falciparum can protect against malaria, probably by destroying the infectious agent as such or the host cells in which they reside, and also by inhibiting a function of the infectious agent critical for its survival. In naturally acquired immunity, all the arms of the immune system are probably activated against all stages of the parasite cycle.
Humoral responses against malarial invasion:
To generalize, three important responses are thought to be very critical in governing the progress of the initial infection. These are also marked as the three most important responses directed against the parasite. These include antibodies directed against parasite proteins expressed on the surface of erythrocytes that prevent sequestration in the microcirculation (Patrick Duffy et al 2001 ), antibodies directed against parasite proteins expressed on the surface of merozoites that prevent invasion of erythrocytes (Sim et al 2001), and antibodies expressed against either type of parasite protein that are capable of mediating antibody dependent cellular inhibition (Bouharoun-Tayoun et al 1995), where biologically active molecules including cytokines, nitric oxide and free oxygen intermediates are released from the reticuloendothelial or other cells after activation through the Fc component of the bound antibody molecule. In addition, biologically active molecules such as cytokines, free oxygen radicals and nitric oxide molecules released from the CD4 T cells after an antigen-specific interaction may also contribute to this immunity. The pathogenesis of the disease may be mediated by these same host-derived biologically active molecules, perhaps elicited by putative malarial toxins released from the infected erythrocytes (Clark et al 2000, Playfair et al 1996 and Schofield et al 2002). Antibodies against these toxins may contribute to naturally acquired immunity. It seems likely that immune responses against sporozoites or infected hepatocytes that limit the number of parasites that emerge from the liver into the bloodstream also play an important role in the resistance to clinical disease demonstrated by individuals with naturally acquired immunity. The most persuasive human data regarding the importance of antibodies against asexual erythrocytic stage antigens come from passive transfer studies in humans (Cohen et al 2001, McGregor et al 1963, and Sabchareon et al 1991).
Cell-Mediated responses against malarial invasion:
T cell mediated immunity plays a very significant role in perpetrating the resistance against the malarial parasite's invasion. For irradiated sporozoite-induced immunity, the protection is thought to be mediated primarily by T cells directed against peptides from parasite proteins expressed in infected hepatocytes by function independent killing via elimination of infected host cells and also by antibodies that reduce sporozoite invasion of hepatocytes (Hoffman et al 1996). Humans immunized with radiation-attenuated sporozoites (Malik et al 1991 and Wizel et al 1995) and also humans naturally exposed to malaria (Aidoo et al 1995, Doolan et al 1997, Flanagan et al 2001, Lalvani et al 1996 and Sedegah et al 1992) have been shown to have CD8+ cytotoxic lymphocytes and CD8+ T cell derived interferon-gamma responses against pre-erythrocytic stage proteins. However data regarding the importance of T cells in the immunity elicited by the radiation attenuated sporozoites come virtually entirely from rodent experiments (Denise et al 1999, Denise et al 2000, Khusmith et al 2001, Romero et al 1989, Schofield et al 1987, Weiss et al 1988, and Weiss et al 1992), in which T cell subsets were depleted or adoptively transferred. These data indicate that in rodent models, protection is always dependent on CD8+ T cells and Interferon gamma, and often dependent on Interleukin-12 and nitric oxide (Denise et al 2000 and Renia et al 1993). Nonetheless, adoptive transfer of antigen specific CD4+ T cells can also protect against sporozoite challenge (Denise et al 2000) and in some strains of mice, depletion of CD4+ T cells also eliminates protection (Wang et al 1996 and Baird et al 1998).
Complexity of the factors governing Immune responses and disease outcomes:
The most well identified difficulty in vaccine research is the rapid alteration of the antigenic determinants. Another area requiring attention is the human genetic polymorphism which is associated with resistance to malaria as in the cases of a and b thalassemia, haemoglobin C, G6PD deficiency, haemoglobin S and haemoglobin E, ABO blood groups (Fya and Fyb) etc.
Human host genetics, transmission dynamics of the parasite, and probably the age of the host contribute to the overwhelming complexity associated with the development of an effective vaccine against malaria. Although it is clear that sickle cell trait protects against severe disease and that other genetic traits may influence outcome, our understanding of the relationship between host genetics and the response to infection is very limited. The elucidation of the sequence of the human genome and the development of scientific tools have led us to a better understanding of the role of host genetic factors in determining the severity of disease associated with infection.
The age of the individual when first exposed to a parasite or a vaccine plays an important role in determining the immune response. A number of reports have suggested that, among non-immune children and non-immune adults, adults are actually more susceptible than children to developing severe disease after their first infection (Baird et al 1995). However, adults acquire immunity faster than children (Kester et al 1998). In addition, the human immune response is also dependent upon transmission dynamics. In areas in which transmission is most intense, infants are at the highest risk of developing severe and fatal malaria, whereas in areas with less intense transmission, older children have a higher incidence of severe and fatal disease than infants.
CURRENT TRENDS AND DEVELOPMENTS IN MALARIAL VACCINE DISCOVERY:
Figure 5. Different types of malarial vaccines, their antigenic composition and essential features.
Vaccine developers around the world are trying to develop all three types of malaria vaccine: pre-erythrocytic, blood stage, and transmission blocking (Figure 5). All three types have been tested in people, and some have shown promise. As noted above, the optimal malaria vaccine will likely combine antigens from all three stages of the malaria parasite's life cycle. Getting there will involve complex studies in the laboratory and in the field to prove that the antigens and the immune responses do not interact in an undesirable manner. Many single-antigen malaria vaccine candidates are being readied for clinical trials, and combination studies are being planned. The deployment of an effective malaria vaccine will take several years, but it will happen.
1. Optimization of Immune responses elicited by Single Stage Targeted vaccines:
As of now, malarial vaccine development is being pursued by three different methods. The most work has been done and progress achieved in attempts to maximize the magnitude and quality of immune responses to a single or a few key antigens, such as the Circumsporozoite protein (CSP), which is a very dominant antigen on the surface of sporozoites and Merozoite Surface Protein (MSP), by immunizing with synthetic peptides or recombinant proteins in an adjuvant. These vaccines are primarily designed to induce antibody and CD4+ responses, but there is also interest in eliciting CD8+ T cell responses.
A. Vaccines targeting Pre-erythrocytic Schizogony:
Extensive work and studies have been performed on the Circumsporozoite protein based recombinant vaccine RTS, S/ASO2. This vaccine is formulated with an oil-in-water emulsion and the immunostimulants monophosphoryl lipid A and the saponin derivative QS21. It is consistently found to protect 40-50% of the volunteers experimentally challenged 2-3 weeks after their last immunization (Stoute et al 1997, Stoute et al 1998 and Bojang et al 2000). When five protected volunteers were challenged six months after the last immunization, only one of the five was protected (Bojang et al 2000). The very first field trials of this vaccine have been conducted in The Gambia by the Medical Research Council, UK. Vaccine efficacy was found to be 71% during the first 9 weeks of surveillance, but subsequently declined in the last 6 weeks (Nardin et al 2000). The first trial of RTS, S/AS02 in a pediatric population was also conducted in the same country. Efforts aimed at improving the vaccine are ongoing and these efforts primarily include the evaluation of alternative adjuvants as well as the combination of RTS, S with other pre-erythrocytic and erythrocytic stage antigens.
New technologies in vaccine science have brought about a huge revolution in new discoveries. One of the most intriguing developments in this field has been that of the Malarial Multiple Antigen Peptide vaccine. This vaccine contains CSP repeat epitopes, which is known to elicit high levels of parasite-specific antibodies and CD4+ T cells in volunteers with specific Human Leukocyte Antigen genotypes (Nardin et al 1993). It has also been demonstrated that a synthetic triepitope polyoxime malaria vaccine containing B cell epitopes and a universal T cell epitope of Plasmodium falciparum CSP has immunogenicity (Alonso et al 1994). In an open labeled phase 1 study, volunteers of diverse HLA types developed antirepeat antibodies and T cells specific for the universal T cell epitope (Alonso et al 1994). A carboxy terminal synthetic peptide of Plasmodium falciparum CSP has also been developed and is under clinical trials.
B. Vaccines targeting Erythrocytic Schizogony:
Vaccines which target the erythrocytic stages of the malarial parasite's lifecycle obstruct the asexual stage of the parasite. SPf66 is one vaccine formulation developed as a polymer containing an 11-amino acid sequence from the N terminus of Plasmodium falciparum Merozoite Surface Protein, the 4-aminoacid repeat of Plasmodium falciparum Circumsporozoite protein, and 2 other short amino acid sequences thought to be from Plasmodium falciparum. Initial trials have demonstrated moderate efficacy in adults and children aged one to five years (Velero et al 1993 and Genton et al 2002).
In yet another discovery, newer vaccines such as the one containing purified recombinant proteins based on three blood stage Plasmodium falciparum proteins: a fragment of Merozoite Surface Protein 1, Merozoite Surface Protein 2, and a portion of the Ring-infected Erythrocyte Surface Antigen, has been formulated in the adjuvant Montanide ISA 720. This vaccine has been shown to have high T cell and antibody induction rates (Graves et al 1988). Vaccines based on the sole Plasmodium falciparum apical membrane antigen-1 are also under developmental studies and assessment.
C. Sexual stage vaccines:
Some of the merozoites in the blood transform into sexual forms, called as gametocytes. These appear in the peripheral blood after 7-10 days of the infection in P. vivax and 10-20 days in P. falciparum infection. When anopheles mosquito bites an infected individual, these gametocytes enter the mosquito and continue their sexual phase of development within the gut wall of the mosquito. This completes the asexual - sexual cycle of the malarial parasite. Vaccines targeting the sexual stages are usually transmission blocking vaccines based on antigens like Pfs25 and Pvs25.
Currently, there are three target antigens of transmission-blocking immunity (i) prefertilization antigens expressed either completely or predominantly in gametocytes, (ii) postfertilization antigens expressed solely or predominantly on zygotes or ookinetes, and (iii) late-midgut-stage antigens such as parasite-produced chitinase required for the ookin