Author: Aris Baras
Institution: Duke University
Date: December 2006
Abstract
The emergence of antibodies as immunotherapeutic agents has offered a tremendous new potential for the treatment of a multitude of human illnesses and diseases. The use of antibodies offers target specificity unparalleled in conventional treatments and therapies. Generation of monoclonal antibodies was made possible by the development of hybridoma technology. Subsequent advances in the development of phage and transgenic mouse technologies have allowed the generation of fully humanized antibodies. Antibody based therapies have used naked antibodies as therapeutic agents or conjugated them with other therapeutic agents such as toxins, radionuclides, and enzymes. Furthermore, antibodies have been increasingly tested for their potential as delivery vehicles for other targeted therapies such as gene therapy. In addition, the innovative designs of antibody fragments are now being tested for their therapeutic efficacy in overcoming some of the issues that have remained as obstacles in whole antibody therapy, such as lack of penetrance and localization. Despite limitations, these therapies have already proven effective in treating allergies, asthma, arthritis, cancer, and a host of other diseases and illnesses. Future advances promise to improve their efficacy.
Introduction
Many antibody-based therapies harness the immune system's own natural defenses and modify them or strengthen them to achieve a therapeutic response. This approach not only eliminates many of the toxic substances used in other treatments, but also provides a much desired alternative to many current treatment regiments, such as chemotherapy and radiation therapy, that come with debilitating consequences for patients. Moreover, those antibody-based therapies that do incorporate toxic chemicals or radioisotopes do so in an efficient manner, narrowing their administration to only specified targets and limiting the exposure of the rest of the body. This specificity is a key advance in better targeting diseases and improving upon current treatment methods.
Extensive studies have focused on the in vivo mechanisms of gene rearrangement that arm the body's immune system with a potentially unlimited arsenal against almost any foreign substance. These principles have been manipulated by scientists to allow millions of years of evolution to come up with answers for many diseases. Furthermore, by mimicking and modifying the natural in vivo processes of somatic hypermutation and affinity maturation, scientists have been able to enhance and strengthen interactions of the immune system by designing more specific, potent, and complete immune responses.
Mechanisms of Action and Clinical Uses of Antibodies
The use of clinically relevant antibodies encompasses a wide array of capabilities and is continuously growing. In general, there are three loosely defined categories of immunotherapeutic antibodies based on their mechanism of action. One of the largest and most clinically pursued of these categories is the use of antibodies as blocking or neutralizing agents. Antibodies can be used to neutralize and block harmful substances either directly, as in binding bacterial toxins and rendering them ineffective, or indirectly by disregulation of signaling pathways and molecular interactions, as in binding ligands or receptors and disrupting their interactions (Brekke et al. 2003).
Secondly, antibodies can be used as cytotoxic mediators in inducing either the complement pathway, increased phagocytosis, or (ADCC) antibody dependent cell mediated cytotoxicity (Stockwin et al. 2003). Antibody therapies based upon these effector functions utilize the antigen specificity of the antigen binding domains to guide and better target the actions of the immune system. Complement fixation, phagocytosis, and ADCC activation, however, are dependent upon interactions with the Fc fragment, the non antigen binding fragment. Therefore, much work has been spent not only in generating specificity, but also in improving the in vivo physiological effector functions mediated by Fc fragment and Fc receptor interactions, such as increasing binding affinity.
A subclass within this group includes multiple cytotoxic mediators, called immunoconjugates, that have been biochemically modified to generate novel therapeutic mechanisms (Stockwin et al. 2003; Hudson et al. 2001; Waldmann 2003). Antibodies have been tagged with toxins, radionuclides, enzymes, and prodrugs. Diptheria toxin and radioisotopes have been chemically conjugated to antibodies or antibody fragments, offering targeted delivery. Such approaches allow increased dosage over what systemic administration would allow. As a result target killing is much more effective and non-targeted cells are spared. In addition, active enzymes can be delivered to their site of action to carry out their therapeutic activity. This type of treatment could allow for the targeted regulation of molecular signaling pathways that could improve the efficacy of many pathway specific small molecule inhibitor based drugs that usually rely on systemic administration. Furthermore, certain chemotherapeutic agents can be administered as prodrugs in antibody directed enzyme prodrug treatment (ADEPT). The enzyme that cleaves the prodrug to its active form is conjugated to antibodies and administered. After they have homed to the tumor site, the inactive prodrugs are administered and by design only activated at the tumor site (Chowdhury et al. 2003). All of these immunoconjugates offer improved treatment designs not by generating novel therapeutic compounds, but simply by delivering them more efficiently to the site of desired action.
Finally, a third class of therapeutic antibodies act as delivery agents. They have been proposed as the agents of delivery for several genetic therapies including gene silencing through RNA interference, allowing targeted delivery of siRNA through immunoconjugated viral vectors (Sanz et al. 2004). Anti-transferrin antibodies have been conjugated to liposomes that carry therapeutic agents (Hudson et al. 2001). These immunoliposomes bind to the transferring receptors on the tightly packed endothelial cells of the capillaries that make up the blood brain barrier. These receptors normally bind and engulf transferrin receptor proteins which carry iron atoms. This clever delivery system affords a simple and elegant mechanism with which to therapeutically overcome some of the most physiologically challenging obstacles to drug administration, such as the blood brain barrier.
Developmental Methods and Technological Progression
The origination of antibody therapy developed from the realization that immunization could afford immune protection. One of the agents providing this immunity was the humoral branch of the immune response, providing specific antibodies that could be detected in the sera of individuals. Behind this observation lay one of the major genetic discoveries. It was later determined that the genetic loci coding for antibodies is subject to the only known programmed rearrangement process in the genome. The many potential coding segments for the different antibody domains are randomly processed together establishing a vast combinatorial diversity. Functional rearrangements are further modified by intense mutagenesis to improve their specificity. The initial challenge in generating therapeutic antibodies was the production limitations of producing clinically relevant amounts of the desired antibodies in mouse models.
However, with the development of hybridoma technology, it was possible to immortalize antibody producing cell lines by fusion with immortal myeloma cell lines. This then afforded the ability to mass produce selected monoclonal antibodies. However, limitations still remained. Antibody secreting plasma cells were initially isolated from mice. This approach ultimately needed revision, as mouse antibodies generated human anti-mouse antibodies. As a result, their therapeutic effects were blocked as they were rapidly cleared and failed to optimally interact with the human immune system, especially in generating effector responses. This setback led to procedures that aimed at eliminating the immunogenicity of these potentially therapeutic antibodies (Gilliland et al. 1999). Advances in transfection techniques allowed for the generation of chimeric antibodies which expressed human constant regions and only mouse antigen binding domains. These greatly improved efficacy, but still displayed immunogenecity. Further progression led to the development of humanized antibodies that were composed of fully human scaffolds with grafted complementarity determining regions generated from mice. These progressions allowed for only the use of the smallest portions of the antigen binding regions of the mouse antibodies, severely reducing their immunogenicity.
The most modern advances have incorporated the use of phage display libraries and the use of transgenic mice in allowing for the production of fully human antibodies (Brekke 2003; Green 1999; Vaswani 1998). In addition, with the discovery of somatic hypermutation and the enzyme responsible for its effects, activation-induced cytodine deaminase, methods have been used to mimic the in vivo process of affinity maturation (Irving et al. 2001). To do so, mutagenesis of selected antibody coding sequences has been used to select for higher specificities and avidities, thus improving efficacy (Chowdhury et al. 2005; Presta et al. 2002).
The New Wave of Engineered Antibodies: Antibody Fragments
Current efforts in the development of therapeutic antibodies have relied upon innovations in designing artificial antibody fragments that isolate only certain domains or segments of whole antibodies (Holliger et al. 2005). For instance, antigen binding fragments have been isolated and used as more effective blocking agents, for they do not induce any effector functions or engage in any other Fc mediated interactions that might compromise their ability to freely circulate. The designs of such innovative strategies have arisen in response to many of the obstacles encountered with the use of whole antibodies as therapeutic agents. Whole antibodies certainly have been successful and will continue to be so in the future. However, certain illnesses provide challenges which are better suited to some of the capabilities that antibody fragments can offer.
Title: Increased tumor killing in multivalent fragment designs Caption: PET scans showing tumor volume indicate the increased tumor killing displayed by multivalent diabodies in comparison to other multivalent antibody based therapies (minibodies and single chain fragments). Reference: Holliger et al. 2005
One of these issues has been avidity. Some of the lack of efficacy for certain treatments has been a displayed lack of avidity for the target immunogen. Despite efforts to artificially mimic affinity maturation, some potential treatments still seem to lack the desired avidity. Innovative designs have begun to polymerize antigen binding fragments, thus increasing their avidity in vivo and subsequently improving their therapeutic efficacy (Chowdhury et al. 2005). These multivalent designs have also been conjugated to many of the therapeutic elements already discussed. Preclinical studies in tumor models how shown improved efficacy of these multivalent fragments in comparison to other antibody fragment based approaches (see Figures 1 and 2) (Holliger et al. 2005).
Title: Comparison of Penetrance through Blood Levels Caption: Graph showing the increased penetrance (higher tumor/blood ratio) of antibody fragments (diabodies and minibodies) and specifically the effectiveness of multivalent fragments to infiltrate tumors in comparison to single chain fragments and whole immunoglobulins. Reference: Holliger et al. 2005
The other main obstacle posed to antibody based therapy has been their limited ability to display any degree of significant penetrance. In general, whole intact antibodies have shown poor localization to certain physiologically guarded areas. These issues of biodistribution have spurred much of the interest in designing antibody fragments. For most therapeutic designs this is not much of an issue. However, this has become a major obstacle in treating solid tumor masses. For this reason, many of the antibody based drugs available are mainly tailored towards treating metastatic cancer and have proven to be quite ineffective with solid tumors. Therefore, efforts have focused on reducing the size of the antibody in order to gain a higher degree of tumor penetrance. Rather than isolate the double chained antibody fragment, scientists have been able to isolate single chain variable region fragments (Sanz et al. 2004). Although these have proven to increase penetrance in preclinical trials, such measures suffer from a lack of avidity and experience more rapid clearance. However, certain measures have been able to counteract this. Polyethylene glycosilation (PEG) of antibody fragments has shown to increase their persistence in the peripheral blood compartment (see Figure 3) while maintaining some of the therapeutic advantages gained by using an antibody fragment (Chapman et al. 1999). Ultimately as with any therapeutic design, there are tradeoffs that must be weighed in order to determine the most efficacious designs.
Title: Comparison of Persistance through Blood Levels Caption: Graph showing the increased persistence (higher % of blood levels) of antibody fragments with the addition of each PEG molecule. Reference: Chapman et al. 1999
On the other hand, where as rapid clearance may be a negative consequence of the use of antibody fragments, it may be useful for certain treatment models. For instance, when using radioimmunoconjugates, agents that display rapid clearance have been considered more beneficial as they limit the amount that non targeted cells are exposed to the radionuclides. Although the use of antibody fragments has not been optimized, it is encouraging to see that they offer many viable options and alternatives to overcome some of the situations in which traditional antibody based therapies have experienced obstacles.
Current Antibody Based Therapies
Due to their high specificity and due to the ability to generate a seemingly unlimited diversity of specificity, antibody based therapies can be used to target many different illnesses and diseases. One of the most successful uses has been in the treatment of allergies and asthma by the use of anti-IgE antibodies that target the Fc fragment of IgE molecules thus disrupting their interactions with tissue mast cell Fc receptors. These interactions normally result in the release of many pharmacologic factors such as histamine, which acts vasoactively, and others that induce smooth muscle contraction. These lead to the asthmatic and allergic symptoms that can be debilitating to much of the population. This treatment proved to be a tremendous success. As can be seen in Figure 4, anti-IgE antibodies have been successful in significantly lowering levels of free IgE (Milgrom et al. 1999).
Title: Effect of anti-IgE Antibodies on Free IgE levels Caption: Anti-IgE antibodies greatly reduce the amount of free IgE molecules that can bind Fc receptors on tissue mast cells Reference: Milgrom et al. 1999
Similar successes have been seen with arthritic treatments based upon antibody therapies (Simon et al. 1999). Much of the focus for rheumatoid arthritis has been blocking the inflammatory effects of the tumor necrosis alpha (TNF-) cytokine (Brekke et al. 2003). Upon binding to its receptor this powerful inflammatory cytokine induces a local inflammatory response that leads to much of the discomfort and symptoms associated with rheumatoid arthritis.
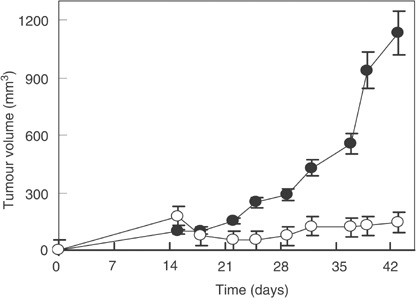
Another group of blocking antibodies targets the epithelial growth factor receptor (EGFR) which is overexpressed on many cancer cells. One of the most promising approaches to attacking cancer has been to focus on inhibiting tumor angiogenesis. Although such treatments aren't as effective in eliminating entire tumor burdens, they offer a powerful manner with which to contain cancers and sensitize them to other chemotherapeutic agents. Figure 5 shows the effect of anti-ErbB2 antibodies in inhibiting tumor growth (Lorenzo et al. 2004). Indeed the trend has been to use such treatments in combination with other agents whose effects are greatly increased by these anti-angiogenic factors. The EGF receptor has been the target of a popular new drug, Herceptin, that has improved treatment of many cancers exhibiting EGFR overexpression.
Title: Effect of anti-EGFR Antibodies in sustaining tumor Caption: Anti-EGFR treated tumor cells (white circles) display a much more contained growth charactersitic in comparison to normal untreated tumor cells Reference: Lorenzo et al. 2004
With regards to cancer, some of the most promising results have been recently achieved by a monoclonal antibody based treatment. A few years ago, a group at the National Cancer Institute at NIH developed a novel recombinant immunotxin targeted at the CD-22 surface marker on hairy cell leukemia cells. The results of their clinical trials were quite astounding. In a trial of 16 patients, 11 experienced complete remissions and 2 experienced partial responses (Kreitman et al. 2001).
Limitations and Potentialities
One of the main limiting factors hindering the efficacy of antibody based therapies is that they still induce allotypic responses (Vaswani et al. 1999). Although the use of fully humanized antibodies has come a long way in reducing immunogenicity, the loci used to develop and produce a certain drug may result in antibodies that induce allotypic immune responses in patients. Such results would recognize the antibodies as foreign and attempt to rapidly clear them. This is the same principle that governs transplant rejections. Although those rejections are more pronounced, due to the fact that whole organs are being transferred, the concept is the same. There is really no complete solution besides developing these drugs on an individual basis, but that is not a feasible alternative at this point. However, it may be the case that companies may have to develop a few different variants of the same drug. For instance, if there are a few common variants in the population, a certain drug may be produced in 5 different forms to accommodate these variances.
One of the major limitations to antibody based therapy is that it relies upon a monoclonal response. Although antibody based immunotherapies aim to harness the immune system's specificity, these therapies only use a fraction of a response typically seen in vivo. Natural immune responses respond in a polyclonal and polyeffector manner. That is, in response to an immunogen, the immune system will provide a response displaying specificity to multiple epitopes on the immunogen and also display multiple effector responses against the immunogen (Gura 2002). This is a much more effective strategy for it targets multiple epitopes and thus reduces the risk of resistance to therapy. Efforts now are being focused on developing mutlivalent antibodies and fragments that recognize different epitopes. In the future, a trend toward the use of polyclonal treatments would undoubtedly improve the efficacy of antibody based therapies.
Another shortcoming of traditional antibody based therapies is that they elicit no T cell help. Furthermore, since they don't not activate or incorporate any other players in the immune response, no sustained responses can be seen as in the case of adoptive cell transfer therapies where whole T cell populations are administered, persist, and sustain a response. With antibody therapies, constant readministration is needed.
Current efforts to overcome these difficulties focus on ways to recruit T cell help and cell mediated killing. Bispecific antibodies have been posed as a solution by using multivalent antibodies that recognize both the target antigen and also bind to T cells, thus recruiting them to the target site. These types of innovations that aim to incorporate more aspects of the immune system ultimately promise to provide much more effective responses.
Discussion
The specificity afforded by antibody based immunotherapies has revolutionized much of the way certain treatments are approached. In many instances, much of the hard work and discovery had been in place and antibodies merely allowed efficient and targeted delivery. The role of antibodies as therapeutic, in themselves, and as agents of delivery for other treatments has overcome many obstacles and offered many avenues with which to approach many illness and diseases that have been difficult to gain access to or treat effectively. However, difficulties still remain. Mainly, future directions aim to include more elements of the host immune response and to include more targets in therapy designs.
Ultimately, however, antibody based therapies depend upon a target. The limitations of these approaches lie in the fact that many targets have not yet been determined for various diseases. In some instances, targets are there, but at very low levels. The use of troybodies has been a way to get around this. These innovative designs display antigen specificity and are coated with antigenic determinants so as to increase the effective amount of antigen once they bind to the target (Hudson et al. 2001). Subsequent therapies can then be administered accordingly. However, in some instances, the targets are self antigens, such as in cancers that overexpress self antigens. These are inherently difficult to approach as they no longer offer specificity. Further work needs to be done to generate more targets through characterization studies and microarray analysis. This sort of target validation will ultimately improve the efficacy of antibody based immunotherapeutics.
References
Brekke, O. H. et al. (2003) Therapeutic Antibodies for Human Diseases at the Dawn of the Twenty-First Century. Nature Reviews Drug Discovery 62, 52-62.
Chapman, A. P. et al. (1999) Therapeutic antibody fragments with prolonged in vivo half-lives. Nature Biotechnology 17, 780-783.
Chowdhury, P. S. et al. (2005) Tailor-made antibody therapeutics. Methods 36, 11-24.
Gilliland, L. K. et al. (1999) Elimination of the Immunogenicity of Therapeutic Antibodies. The Journal Immunology 64, 3664-3671.
Green, L. L. (1999) Antibody engineering via genetic engineering of the mouse: XenoMouse strains are a vehicle for the facile generation of therapeutic human monoclonal antibodies. Journal of Immunological Methods 231, 11-23.
Gura, T. (2002) Magic bullets hit the target. Nature 417, 584-586.
Holliger, P. et al. (2005) Engineered Antibody Fragments and the Rise of Single Domains. Nature Biotechnology 23(9), 1126-1136.
Hudson, P. J. et al. (2001) Engineered antibodies. Nature Medecine 9, 129-134.
Irving, R. A. et al. (2001) Ribosome display and affinity maturation: from antibodies to single V-domains and steps towards cancer therapeutics. Journal of Immunological Methods 248, 31-45.
Kreitman, R. J. et al. (2001) Efficacy of the Anti-CD22 Recombinant Immunotoxin BL22 in Chemotherapy-Resistant Hairy Cell Leukemia. NEJM.345, 241-247.
Lorenzo, C. D. et al. (2004) A human, compact, fully functional anti-ErbB2 antibody as a novel antitumour agent. British Journal of Cancer 91, 1200-1204.
Milgrom, H. et al. (1999) Treatment of Allergic Asthma with Monoclonal Anti-IgE Antibody. NEJM 341, 1966-1973.
Presta, L. G. et al. (2002) Celebrating Diversity and Mimicking Maturation. Nature
Biotechnology 20, 1096-1097.
Sanz, L. et al. (2004) Antibodies and gene therapy: teaching old magic bullets' new tricks. Trends in Immunology 25, 86-91.
Simon, L. S. et al. (2000) New and Future Therapies for Rheumatoid Arthritis. Rheumatology 39, 36-42.
Stockwin, L. H. et al. (2003) The role of therapeutic antibodies in drug discovery. Biochemical Society Transactions 31, 434-436.
Vaswani, S. K. et al. (1998) Humanized antibodies as potential therapeutic drugs. Annals of Allergy, Asthma, and Immunology 64, 105-119.
Waldmann, T. A. (2003) Immunotherapy: past, present and future. Nature Medecine 9, 269-277.