Author: Dabkowska Aleksandra
Institution: Biochemistry & Environmental Science
Date: June 2005
Bullseye.
Bullseye pattern on petri dish of E.coli as viewed by fluorescence microscope. The receiver cells form a red and green, bullseye-coloured colony around a blue disk in the middle that contains the sender cells. Image courtesy of Dr. Ron Weiss.
That is the pattern that you would see if you looked onto the lawn of E.coli covering the petri dish from the labs of Frances Arnold and Ron Weiss. What is actually going on between the cells on the petri dish is even stranger: the bacteria are "telling" each other to form the bullseye. These bacterial cells were successfully programmed to communicate with each other in order to form a color pattern specified by the researcher. In effect, the bacteria have been made into microscopic robots.
"The basic idea that we're trying to demonstrate," says Frances Arnold, a professor of chemical engineering at Caltech, who published the results in the April 28th issue of Nature, "is that by having programmed gene expression, these cells can perform a non-natural function."
Subhayu Basu, Yoram Gerchman, Cynthia H. Collins, Frances Arnold and Ron Weiss from Princeton and Caltech have built, what is in Arnold's words, a "simple but complex circuitry" that allows one type of cells (the senders) to biologically sense a chemical in the environment and relay the message onto other cells (the receivers). The receiver cells then respond to the signal by manufacturing proteins that fluoresce red. Different patterns can be induced by simply placing the sender cells in different positions.
The researchers also created mathematical models that they used to predict how the bullseye pattern would form on the initially colourless lawn of E.coli under different conditions.
"The mathematical model tells us what kind of behaviour is possible, and what range of parameters gives rise to specific behaviours," says Arnold. The modelling helped the researchers choose the right alignment of sender and receiver cells by showing how certain changes in kinetic parameters affect the pattern formation.
Weiss and Arnold have transformed E.coli cells into an artificial multi-cellular system that is like a circuit. When the sender cell is switched on, it sends a signal to the next cell, also turning it on like a light bulb. However, instead of using electrical energy to turn on light bulbs, these cells communicate using secreted signalling molecules that activate a reporter gene.
It turns out that bacterial cells secrete signalling molecules to communicate with each other all the time in order to form communities that can survive. In the case of programmable cells, researchers simply harnessed the cells' ability to communicate with other cells using "quorum sensing".
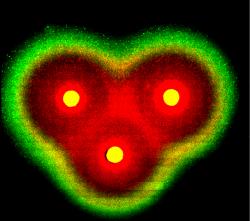
A heart-shaped pattern on petri dish of E.coli as viewed by fluorescence microscope. Using a different number and alignment of sender cells, various patterns were obtained. Image courtesy of Dr. Ron Weiss.
Single cells use quorum sensing to figure out how many other cells are around them. Then, if they detect changes in the concentration of nutrients or poisons in their vicinity, the bacterial community can coordinate to avoid the danger. It is like many of our disaster warning systems, but instead of using the telephone, radio or conversation, bacteria "talk" by releasing small molecules into their environment.
Christopher Tang, a researcher at the Centre for Molecular Microbiology and Infection at Imperial College in London, investigates bacterial communication in pathogens. He has recently studied a quorum-sensing system that uses signal molecules produced by a gene called LuxS. Since LuxS is also involved in metabolism and the pathway is found in over 55 species, Tang wonders whether it might be a universal language for interspecies communication.
Tang says there are many reasons why bacteria would want to communicate with each other. Pathogens, for instance, may want to postpone their invasion of the host until their attack is strengthened by large numbers.
"Researchers are looking to inhibit bacterial cross-talk in bacteria.. which could lead to a paradigm shift in terms of treatment, " explains Tang. Imagine, instead of taking an antibiotic to kill bacteria once the infection is in full swing, you could be "scrambling" the communication between bacteria, before they are able to prepare their attack.
Being able to control pathogens by using their own communication against them is just one way that researchers can apply their understanding of communication between cells. Although no specific game-plans are drawn up yet, researchers are getting closer to fulfilling the need for controllable cellular circuits which can be used to engineer replacement tissue or biomaterials.
Synthetic biologists build systems that are inspired by nature. Sometimes the goal is simply to re-engineer cellular machinery to produce large amounts of some useful compound. Other times, synthetic engineering helps scientists discover how nature works by re-building it from the ground up. The members of the new field of synthetic biology come from many different backgrounds, and the various combinations often make each project new territory. But that only makes it more interesting from the standpoint of a researcher like Frances Arnold.
"When you're working in a field [such as synthetic biology] that is wide open", reflects Arnold, "You get to define what the problems are. If you are flexible and fast, you get the chance to create."
Inspired by the flexible and fast natural world, the rational design of programmable cells is a technological endeavour that can provide tiny living machines targeted for use to future social and economic benefit. In creating these cellular circuits, Arnold, Weiss, and colleagues in the field of synthetic biology seem to have hit the bull's eye.
Further Reading
Bassler BL. (1999). How bacteria talk to each other: regulation of gene expression by quorum sensing. Curr Opin Microbiol. 2:582587.
Basu S, Gerchman Y, Collins CH, et al. (2005). A synthetic multicellular system for programmed pattern formation. Nature. 434:1130-4.
Henke JM and BL Bassler. (2004). Bacterial social engagements. Trends Cell Biol. 14:648-56.
Vendeville A, Winzer K, Heurlier K, et al. (2005). Making 'sense' of metabolism: autoinducer-2, LuxS and pathogenic bacteria. Nat Rev Microbiol. 3:383-96.